Progestogen-releasing intrauterine systems for heavy menstrual bleeding
- PMID: 32529637
- PMCID: PMC7388184
- DOI: 10.1002/14651858.CD002126.pub4
Progestogen-releasing intrauterine systems for heavy menstrual bleeding
Abstract
Background: Heavy menstrual bleeding (HMB) impacts the quality of life of otherwise healthy women. The perception of HMB is subjective and management depends upon, among other factors, the severity of the symptoms, a woman's age, her wish to get pregnant, and the presence of other pathologies. Heavy menstrual bleeding was classically defined as greater than or equal to 80 mL of blood loss per menstrual cycle. Currently the definition is based on the woman's perception of excessive bleeding which is affecting her quality of life. The intrauterine device was originally developed as a contraceptive but the addition of progestogens to these devices resulted in a large reduction in menstrual blood loss: users of the levonorgestrel-releasing intrauterine system (LNG-IUS) reported reductions of up to 90%. Insertion may, however, be regarded as invasive by some women, which affects its acceptability.
Objectives: To determine the effectiveness, acceptability and safety of progestogen-releasing intrauterine devices in reducing heavy menstrual bleeding.
Search methods: We searched the Cochrane Gynaecology and Fertility Specialised Register, CENTRAL, MEDLINE, Embase, PsycINFO and CINAHL (from inception to June 2019); and we searched grey literature and for unpublished trials in trial registers.
Selection criteria: We included randomised controlled trials (RCTs) in women of reproductive age treated with LNG-IUS devices versus no treatment, placebo, or other medical or surgical therapy for heavy menstrual bleeding.
Data collection and analysis: Two authors independently extracted data, assessed risk of bias and conducted GRADE assessments of the certainty of evidence.
Main results: We included 25 RCTs (2511 women). Limitations in the evidence included risk of attrition bias and low numbers of participants. The studies compared the following interventions. LNG-IUS versus other medical therapy The other medical therapies were norethisterone acetate, medroxyprogesterone acetate, oral contraceptive pill, mefenamic acid, tranexamic acid or usual medical treatment (where participants could choose the oral treatment that was most suitable). The LNG-IUS may improve HMB, lowering menstrual blood loss according to the alkaline haematin method (mean difference (MD) 66.91 mL, 95% confidence interval (CI) 42.61 to 91.20; 2 studies, 170 women; low-certainty evidence); and the Pictorial Bleeding Assessment Chart (MD 55.05, 95% CI 27.83 to 82.28; 3 studies, 335 women; low-certainty evidence). We are uncertain whether the LNG-IUS may have any effect on women's satisfaction up to one year (RR 1.28, 95% CI 1.01 to 1.63; 3 studies, 141 women; I² = 0%, very low-certainty evidence). The LNG-IUS probably leads to slightly higher quality of life measured with the SF-36 compared with other medical therapy if (MD 2.90, 95% CI 0.06 to 5.74; 1 study: 571 women; moderate-certainty evidence) or with the Menorrhagia Multi-Attribute Scale (MD 13.40, 95% CI 9.89 to 16.91; 1 trial, 571 women; moderate-certainty evidence). The LNG-IUS and other medical therapies probably give rise to similar numbers of women with serious adverse events (RR 0.91, 95% CI 0.63 to 1.30; 1 study, 571 women; moderate-certainty evidence). Women using other medical therapy are probably more likely to withdraw from treatment for any reason (RR 0.49, 95% CI 0.39 to 0.60; 1 study, 571 women, moderate-certainty evidence) and to experience treatment failure than women with LNG-IUS (RR 0.34, 95% CI 0.26 to 0.44; 6 studies, 535 women; moderate-certainty evidence). LNG-IUS versus endometrial resection or ablation (EA) Bleeding outcome results are inconsistent. We are uncertain of the effect of the LNG-IUS compared to EA on rates of amenorrhoea (RR 1.21, 95% CI 0.85 to 1.72; 8 studies, 431 women; I² = 21%; low-certainty evidence) and hypomenorrhoea (RR 0.98, 95% CI 0.73 to 1.33; 4 studies, 200 women; low-certainty evidence) and eumenorrhoea (RR 0.55, 95% CI 0.30 to 1.00; 3 studies, 160 women; very low-certainty evidence). We are uncertain whether both treatments may have similar rates of satisfaction with treatment at 12 months (RR 0.95, 95% CI 0.85 to 1.07; 5 studies, 317 women; low-certainty evidence). We are uncertain if the LNG-IUS compared to EA has any effect on quality of life, measured with SF-36 (MD -14.40, 95% CI -22.63 to -6.17; 1 study, 33 women; very low-certainty evidence). Women with the LNG-IUS compared with EA are probably more likely to have any adverse event (RR 2.06, 95% CI 1.44 to 2.94; 3 studies, 201 women; moderate-certainty evidence). Women with the LNG-IUS may experience more treatment failure compared to EA at one year follow up (persistent HMB or requirement of additional treatment) (RR 1.78, 95% CI 1.09 to 2.90; 5 studies, 320 women; low-certainty evidence); or requirement of hysterectomy may be higher at one year follow up (RR 2.56, 95% CI 1.48 to 4.42; 3 studies, 400 women; low-certainty evidence). LNG-IUS versus hysterectomy We are uncertain whether the LNG-IUS has any effect on HMB compared with hysterectomy (RR for amenorrhoea 0.52, 95% CI 0.39 to 0.70; 1 study, 75 women; very low-certainty evidence). We are uncertain whether there is difference between LNG-IUS and hysterectomy in satisfaction at five years (RR 1.01, 95% CI 0.94 to 1.08; 1 study, 232 women; low-certainty evidence) and quality of life (SF-36 MD 2.20, 95% CI -2.93 to 7.33; 1 study, 221 women; low-certainty evidence). Women in the LNG-IUS group may be more likely to have treatment failure requiring hysterectomy for HMB at 1-year follow-up compared to the hysterectomy group (RR 48.18, 95% CI 2.96 to 783.22; 1 study, 236 women; low-certainty evidence). None of the studies reported cost data suitable for meta-analysis.
Authors' conclusions: The LNG-IUS may improve HMB and quality of life compared to other medical therapy; the LNG-IUS is probably similar for HMB compared to endometrial destruction techniques; and we are uncertain if it is better or worse than hysterectomy. The LNG-IUS probably has similar serious adverse events to other medical therapy and it is more likely to have any adverse events than EA.
Copyright © 2020 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Conflict of interest statement
AL, MB and VJ have no conflict of interest to declare.
Figures
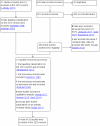

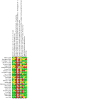




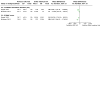






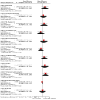






















Update of
-
Progesterone or progestogen-releasing intrauterine systems for heavy menstrual bleeding.Cochrane Database Syst Rev. 2015 Apr 30;(4):CD002126. doi: 10.1002/14651858.CD002126.pub3. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2020 Jun 12;6:CD002126. doi: 10.1002/14651858.CD002126.pub4. PMID: 25924648 Updated.
Comment in
-
Levonorgestrel-Releasing Intrauterine System for Reducing Heavy Menstrual Bleeding.Am Fam Physician. 2021 Aug 1;104(2):138-139. Am Fam Physician. 2021. PMID: 34383439 No abstract available.
References
References to studies included in this review
Ashraf 2017 {published data only}
-
- Ashraf M, Habib-Ur-Rehman, A, Shehzad, Z, AlSharari S, Murtaza G. Clinical efficacy of levonorgestrel and norethisterone for the treatment of chronic abnormal uterine bleeding. Journal of the Pakistan Medical Association 2017;67(9):1331-8. [28924270] - PubMed
-
- Ashraf M, Murtaza G. Levonorgestrel intrauterine system and norethisterone for the treatment of chronic abnormal uterine bleeding: A randomized clinical comparison in the patients of Bahawalpur, Pakistan. Value in Health 2016;19(7):A577. [ 613235950]
Barrington 2003 {published data only}
-
- Barrington JW, Arunkalaivanan AS, Abdel-Fattah M. Comparison between the levonorgestrel intrauterine system (LNG-IUS) and thermal balloon ablation in the treatment of menorrhagia. European Journal of Obstetrics, Gynecology, and Reproductive Biology 2003;108(1):72-4. - PubMed
Crosignani 1997 {published data only}
-
- Crosignani PG, Vercellini P, Mosconi P, Oldani S, Cortesi I, De Giorgi O. Levonorgestrel-releasing intrauterine device versus hysteroscopic endometrial resection in treatment of dysfunctional uterine bleeding. Obstetrics and Gynecology Clinics of North America 1997;90(2):257-63. - PubMed
de Souza 2010 {published data only}
-
- Silva-Filho AL, Pereira FAN, Souza SS, Loures LF, Rocha APC, Valadares CN, et al. Five-year follow-up of levonorgestrel-releasing intrauterine system versus thermal balloon ablation for the treatment of heavy menstrual bleeding: a randomised controlled trial. Contraception 2013;87(4):409-15. - PubMed
-
- Souza SS, Camargos AF, Rezende CP, Pereira FAN, Araujo CAA, Filho ALS. A randomised prospective trial comparing the levonorgestrel-releasing intrauterine system with thermal balloon ablation for the treatment of heavy menstrual bleeding. Contraception 2010;81(3):226-31. - PubMed
Endrikat 2009 {published data only}
-
- Endrikat J, Shapiro H, Lukkari-Lax E, Kunz M, Schmidt W, Fortier M. A Canadian, multicentre study comparing efficacy of a levonorgestrel-releasing intrauterine system to an oral contraceptive in women with idiopathic menorrhagia. Journal d'Obstetrique et Gynecologie du Canada : JOGC [Journal of Obstetrics and Gynaecology Canada : JOGC] 2009;31(4):340-7. - PubMed
Ergun 2012 {published data only}
-
- Ergun B, Bastu E, Kuru O, Sen S, Kilic Y, Dural O. Comparison of rollerball endometrial ablation and levonorgestrel releasing intrauterine system in the management of abnormal uterine bleeding. International Journal of Gynaecology and Obstetrics 2012;119(S3):S672.
-
- Ergun B, Kuru O, Sen S, Kilic Y, Bastu E. Rollerball endometrial ablation versus levonorgestrel releasing intrauterine system in the management of abnormal uterine bleeding. Ginecologia.ro 2012;8(30):199-201.
Ghazizadeh 2014 {published data only}
-
- Ghazizadeh S, Panahi Z, Ghanbari Z, Menshadi AT, Farahmandian T, Javadian P. Comparative efficacy of Novasure, the levonorgestrel-releasing intrauterine system and hysteroscopic endometrial resection in the treatment of menorrhagia: a randomised clinical trial. Journal of Gynecologic Surgery 2014;30(4):215-8.
Gupta 2013 {published data only}
-
- Gupta J, Kai J, Middleton L, Pattison H, Gray R, Daniels J. Levonorgestrel intrauterine system versus medical therapy for menorrhagia. New England Journal of Medicine 2013;368(2):128-37. - PubMed
-
- Gupta JK, Daniels JP, Middleton LJ, Pattison HM, Prileszky G, Roberts TE, et cols. A randomised controlled trial of the clinical effectiveness and cost-effectiveness of the levonorgestrel-releasing intrauterine system in primary care against standard treatment for menorrhagia: The ECLIPSE trial. Health technology Assessment 2015;19(88):1-118. [DOI: 10.3310/hta19880] - DOI - PMC - PubMed
-
- Kai J, Middleton L, Daniels J, Tryposkiadis K, Pattison H, Gupta J. Usual medical treatments or levonorgestrel-IUS for women with heavy menstrual bleeding: long-term randomised pragmatic trial in primary care. British Journal of General Practice 2016;66(653):e861-e870. [DOI: 10.3399/bjgp16X687577] - DOI - PMC - PubMed
-
- Sanghera S, Roberts T, Barton P, Daniels J, Middleton L, Gennard L, et al. Cost effectiveness of levonorgestrel intrauterine system for menorrhagia. BJOG 2013;120:390-1.
Herman 2013 {published data only}
-
- Beelen P, Van Den Brink MJ, Geomini P, Dekker J, Herman M, Duijnhoven R, et al. RCT comparing the LNG-IUS versus endometrial ablation in women with heavy menstrual bleeding (MIRA). International Journal of Gynaecology and Obstetrics 2018;143(3):318-9. [10.1002/ijgo.12582]
Hurskainen 2001 {published data only}
-
- Halmesmaki K, Hurskainen R, Teperi J, Grenman S, Kivela A, Kujansuu E, et al. The effect of hysterectomy or levonorgestrel-releasing intrauterine system on sexual functioning among women with menorrhagia: a 5-year randomised controlled trial. British Journal of Obstetrics and Gynaecology 2007;114(5):563-8. - PubMed
-
- Halmesmaki K, Hurskainen R, Tiitinen A, Teperi J, Grenman S, Kivela A, et al. A randomized controlled trial of hysterectomy or levonorgestrel-releasing intrauterine system in the treatment of menorrhagia - effect on FSH levels and menopausal symptoms. Human Reproduction (Oxford, England) 2004;19(2):378-82. - PubMed
-
- Halmesmaki KH, Hurskainen RA, Paavonen JA, Tuppurainen MT. Randomised controlled trial of the effect of hysterectomy or LNG-IUS use on bone mineral density: a five-year follow up. Therapy 2006;3(4):509-15.
-
- Heliovaara-Peippo S, Halmesmaki K, Hurskainen R, Teperi J, Grenman S, Kivela A, et al. The effect of hysterectomy or levonorgestrel-releasing intrauterine system on lower abdominal pain and back pain among women treated for menorrhagia: a five-year randomised controlled trial. Acta Obstetricia et Gynecologica Scandinavica 2009;88(12):1389-96. - PubMed
-
- Heliovaara-Peippo S, Halmesmaki K, Hurskainen R, Teperi J, Grenman S, Kivela A, et al. The effect of hysterectomy or levonorgestrel-releasing intrauterine system on lower urinary tract symptoms: a 10-year follow-up study of a randomised trial. British Journal of Obstetrics and Gynaecology 2010;117(5):602-9. - PubMed
Irvine 1998 {published data only}
-
- Irvine GA, Campbell-Brown MB, Lumsden MA, Heikkila A, Walker JJ, Cameron IT. Randomised comparative trial of the levonorgestrel intrauterine system and norethisterone for the treatment of idiopathic menorrhagia. British Journal of Obstetrics and Gynaecology 1998;105(6):592-8. - PubMed
Kaunitz 2010 {published data only}
-
- Kaunitz AM, Bissonnette F, Monteiro I, Lukkari-Lax E, DeSanctis Y, Jensen J. Levonorgestrel-releasing intrauterine system for heavy menstrual bleeding improves hemoglobin and ferritin levels. Contraception 2012;86(5):452-7. - PubMed
-
- Kaunitz AM, Bissonnette F, Monteiro I, Lukkari-Lax E, Muysers C, Jensen JT. Levonorgestrel-releasing intrauterine system or medroxyprogesterone for heavy menstrual bleeding. Obstetrics and Gynecology 2010;116(3):625-32. - PubMed
Kilic 2009 {published data only}
-
- Kilic S, Yuksel B, Doganay M, Bardakci H, Akinsu F, Uzunlar O, et al. The effect of levonorgestrel-releasing intrauterine device on menorrhagia in women taking anticoagulant medication after cardiac valve replacement. Contraception 2009;80(2):152-7. - PubMed
Kiseli 2016 {published data only}
-
- Kiseli M, Kayikcioglu F, Evliyaoglu O, Haberal A. Comparison of therapeutic efficacies of norethisterone, tranexamic acid and levonorgestrel-releasing intrauterine system for the treatment of heavy menstrual bleeding: a randomized controlled study. Gynecologic and Obstetric Investigation 2016;81(5):447-53. [CRSSTD: 7929734] - PubMed
Kittelsen 1998 {published data only}
-
- Istre O, Trolle B. Treatment of menorrhagia with the levonorgestrel intrauterine system versus endometrial resection. Fertility and Sterility 2001;76(2):304-9. - PubMed
-
- Kittelsen N, Istre O. A randomized study comparing levonorgestrel intrauterine system (LNG IUS) and transcervical resection of the endometrium (TCRE) in the treatment of menorrhagia: preliminary results. Gynaecological Endoscopy 1998;7(2):61-5.
-
- Rauramo I, Elo I, Istre O. Long-term treatment of menorrhagia with levonorgestrel intrauterine system versus endometrial resection. Obstetrics and Gynecology 2004;104(6):1314-21. - PubMed
Malak 2006 {published data only}
-
- Malak KA, Shawki O. Management of menorrhagia with the levonorgestrel intrauterine system versus endometrial resection. Gynecological Surgery 2006;3(4):275-80.
Ozdegirmenci 2011 {published data only}
-
- Ozdegirmenci O, Kayikcioglu F, Akgul MA, Kaplan M, Karcaaltincaba M, Haberal A, et al. Comparison of levonorgestrel intrauterine system versus hysterectomy on efficacy and quality of life in patients with adenomyosis. Fertility and Sterility 2011;95(2):497-502. - PubMed
Reid 2005a {published data only}
-
- Reid PC, Virtanen-Kari S. Randomised comparative trial of the levonorgestrel intrauterine system and mefenamic acid for the treatment of idiopathic menorrhagia: a multiple analysis using total menstrual fluid loss, menstrual blood loss and pictorial blood loss measurements. British Journal of Obstetrics and Gynaecology 2005;112(8):1121-5. - PubMed
Sayed 2011 {published data only}
-
- Sayed GH, Zakherah MS, El-Nashar SA, Shaaban MM. A randomised clinical trial of a levonorgestrel-releasing intrauterine system and a low-dose combined oral contraceptive for fibroid-related menorrhagia. International Journal of Gynaecology and Obstetrics 2011;112(2):126-30. - PubMed
Sesti 2012 {published data only}
-
- Sesti F, Piancatelli R, Pietropoli A, Ruggeri V, Piccione E. Levonorgestrel-releasing intrauterine system versus laparoscopic supracervical hysterectomy for the treatment of heavy menstrual bleeding: a randomised study. Journal of Women's Health 2012;21(8):851-7. - PubMed
Shabaan 2011 {published data only}
-
- Shabaan MM, Zakherah MS, El-Nashar SA, Sayed GH. Levonorgestrel-releasing intrauterine system compared to low dose combined oral contraceptive pills for idiopathic menorrhagia: a randomised clinical trial. Contraception 2011;83(1):48-54. - PubMed
Shaw 2007 {published data only}
-
- Shaw RW, Symonds IM, Tamizian O, Chaplain J, Mukhopadhyay S. Randomised comparative trial of thermal balloon ablation and levonorgestrel intrauterine system in patients with idiopathic menorrhagia. Australian & New Zealand Journal of Obstetrics & Gynaecology 2007;47(4):335-40. - PubMed
-
- Shaw RW, Symonds IM, Tamizian O. Randomised comparative trial of thermal balloon ablation (TBA) v levonorgestrel intrauterine system (LNG IUS) in idiopathic menorrhagia. In: XVIII FIGO World Congress of Gynecology and Obstetrics. Vol. 4. 2006:119. - PubMed
Soysal 2002 {published data only}
-
- Soysal M, Soysal S, Ozer S. A randomized controlled trial of levonorgestrel releasing IUD and thermal balloon ablation in the treatment of menorrhagia. Zentralblatt fur Gynakologie 2002;124(4):213-9. - PubMed
TALIS 2006 {published data only}
-
- Brown PM, Farquhar CM, Lethaby A, Sadler LC, Johnson NP. Cost-effectiveness analysis of levonorgestrel intrauterine system and thermal balloon ablation for heavy menstrual bleeding. BJOG 2006;113(7):797-803. - PubMed
-
- Busfield RA, Farquhar CM, Sowter MC, Lethaby A, Sprecher M, Yu Y, et al. A randomised trial comparing the levonorgestrel intrauterine system and thermal balloon ablation for heavy menstrual bleeding. BJOG 2006;113(3):257-63. - PubMed
Tam 2006 {published data only}
-
- Tam WH, Yuen PM, Ng DPS, Leung PL, Lok IH, Rogers MS. Health status function after treatment with thermal balloon ablation and levonorgestrel intrauterine system for idiopathic menorrhagia: a randomised study. Gynecologic and Obstetric Investigation 2006;62(2):84-8. - PubMed
References to studies excluded from this review
Abu Hashim 2013 {published data only}
Adiguzel 2017 {published data only}
-
- Adiguzel C, Seyfettinoglu S, Aka Satar D, Arlier S, Eskimez E, Kaya F, Nazik H. Evaluation of quality of life and cost-effectiveness of definitive surgery and the levonorgestrel intrauterine system as treatment options for heavy menstrual bleeding. Turkish Journal of Medical Sciences 2017;47(3):789-94. [DOI: 10.3906/sag-1512-115] - DOI - PubMed
Apter 2016 {published data only (unpublished sought but not used)}
-
- Apter D, Briggs P, Tuppurainen M, Grunert J, Lukkari-Lax E, Rybowski S, Gemzell-Danielsson K. A 12-month multicenter, randomized study comparing the levonorgestrel intrauterine system with the etonogestrel subdermal implant. Fertility and Sterility 2016;106(1):151-7.e5. [DOI: 10.1016/j.fertnstert.2016.02.036] - DOI - PubMed
Buyuktuna 2016 {published data only}
-
- Buyuktuna N, Sumer F. Comparison of mirena intrauterine device with oral progesterone treatment and surgical treatment in idiopathic menorrhagia patients: Results of a cost-minimisation analysis. Value in Health 2016;19(7):A698. [613234471]
Cameron 1987 {published data only}
-
- Cameron IT, Leask R, Kelly RW, Baird DT. The effects of danazol, mefenamic acid, norethisterone and a progesterone-impregnated coil on endometrial prostaglandin concentrations in women with menorrhagia. Prostaglandins 1987;34(1):99-110. - PubMed
-
- Cameron IT. Dysfunctional uterine bleeding. Bailliere's Clinical Obstetrics and Gynaecology 1989;3(2):315-27. - PubMed
Ghazizadeh 2011 {published data only}
-
- Ghazizadeh S, Bakhtiari F, Rahmanpour H, Davari-Tanha F, Ramezanzadeh F. A randomised clinical trial to compare levonorgestrel-releasing intrauterine system (Mirena) vs transcervical endometrial resection for treatment of menorrhagia. International Journal of Women's Health 2011;3:207-11. - PMC - PubMed
Gupta 2006 {published data only}
-
- Gupta B, Mittal S, Misra R, Deka D, Dadhwal V. Levonorgestrel-releasing intrauterine system vs transcervical endometrial resection for dysfunctional uterine bleeding. International Journal of Gynaecology and Obstetrics 2006;95:261-6. - PubMed
Janssen 1999 {published data only}
-
- Janssen CAH. Menorrhagia and the 3-keto-desogestrel-copper medicated intrauterine device. European Journal of Obstetrics, Gynecology, and Reproductive Biology 1999;85:135-6. - PubMed
Karacaoglu 2001 {published data only}
-
- Karacaoglu U, Cebi Z, Savan K, Yasar L, Cankaya A, Kupelioglu L, et al. Comparing hysteroscopic ablation with levonorgestrel-releasing intrauterine device in abnormal uterine bleeding cases. Jinekoloji Ve Obstetrik Dergisi 2001;15(3):172-5.
Karimi‐Zarchi 2013 {published data only}
-
- Karimi-Zarchi M, Dehghani-Firoozabadi R, Tabatabaie A, Dehghani-Firoozabadi Z, Teimoori S, Chiti Z, et al. A comparison of the effect of levonorgestrel IUD with oral medroxyprogesterone acetate on abnormal uterine bleeding with simple endometrial hyperplasia and fertility preservation. Clinical and Experimental Obstetrics & Gynecology 2013;40(3):421-4. - PubMed
Kucuk 2008 {published data only}
-
- Kucuk U, Ertan K. Continuous oral or intramuscular medroxyprogesterone acetate versus the levonorgestrel releasing intrauterine system in the treatment of perimenopausal menorrhagia: a randomised prospective controlled trial in female smokers. Clinical and Experimental Obstetrics & Gynecology 2008;35(1):57-60. - PubMed
Lahteenmaki 1998 {published data only}
Lee 2013 {published data only}
-
- Lee BS, Ling X, Asif S, Kraemer P, Hanisch JU, Inki P. Levonorgestrel-releasing intrauterine system versus conventional medical therapy for heavy menstrual bleeding in the Asia-Pacific region. International Journal of Gynaecology and Obstetrics 2013;121:24-30. - PubMed
-
- Xu L, Lee BS, Asif S, Kraemer P, Inki P. Satisfaction and health-related quality of life in women with heavy menstrual bleeding; results from a non interventional trial of the levonorgestrel-releasing intrauterine system or conventional medical therapy. International Journal of Women's Health 2014;6:547-54. - PMC - PubMed
Mawet 2014 {published data only}
-
- Mawet M, Nollevaux F, Nizet D, Wijzen F, Gordenne V, Tasev N, et al. Impact of a new levonorgestrel intrauterine system, Levosert(), on heavy menstrual bleeding: results of a one-year randomised controlled trial. European Journal of Contraception & Reproductive Health Care 2014;19(3):169-79. [10.3109/13625187.2014.894184] - PMC - PubMed
McMillan 1998 {unpublished data only}
-
- McMillan L. Randomised controlled study comparing the efficacy of Levonorgestrel intrauterine device with Nd:YAG laser endometrial ablation for the treatment of menorrhagia and premenstrual syndrome. National Research Register, id N0080056736 .
McMillan 2005 {published data only}
-
- McMillan L. Randomised controlled study comparing the efficacy of levonorgestrel intrauterine device with surgical treatment of menorrhagia by total endometrial ablation. National Research Register, id N0080056730 .
Milsom 1991 {published data only}
-
- Milsom I, Andersson K, Andersch B, Rybo G. A comparison of flurbiprofen, tranexamic acid and a levonorgestrel-releasing intrauterine contraceptive device in the treatment of idiopathic menorrhagia. American Journal of Obstetrics and Gynecology 1991;164(3):879-83. - PubMed
Rogerson 1999 {unpublished data only}
-
- Rogerson L, Crocombe W, Duffy S. SMART - Satisfaction with Mirena and ablation: a randomised trial (abstract). In: British Society of Gynaecological Endocrinology. 1999.
Romer 2000 {published data only}
-
- Romer T. Prospective comparison study of levonorgestrel IUD versus Roller-Ball endometrial ablation in the management of refractory recurrent hypermenorrhea. European Journal of Obstetrics, Gynecology, and Reproductive Biology 2000;90(1):27-9. - PubMed
Tosun 2014 {published data only}
-
- Tosun AK, Tosun I, Suer N. Comparison of levonorgestrel-releasing intrauterine device with oral progestins in heavy menstrual bleeding (HMB) cases with uterine leiomyoma (LNG-IUD and oral progestin usage in myoma uteri). Pakistan Journal of Medical Sciences Quarterly 2014;30(4):834-9. - PMC - PubMed
References to ongoing studies
SHiPP 2013 {unpublished data only}
-
- SHiPP (Stopping Heavy Periods Project), Identifier NCT02002260. Ongoing study. February 2013 ‒ estimated completion date January 2017. Contact author for more information.
Additional references
Andersson 1994
-
- Andersson K, Odlind V, Rybo G. Levonorgestrel-releasing and copper-releasing (Nova T) IUDs during five years of use: a randomized comparative trial. Contraception 1994;49(1):56-72. - PubMed
Baldwin 2013
-
- Baldwin MK, Jensen JT. Contraception during the perimenopause. Maturitas 2013;76(3):235-42. - PubMed
Bayer S.A. 2019
-
- Bayer SA. Number of countries around the world where Mirena is available. email to: Magdalena Bofill 2019.
Berqvist 1983
-
- Berqvist A, Rybo G. Treatment of menorrhagia with intrauterine release of progesterone. British Journal of Obstetrics and Gynaecology 1983;90(3):255-8. - PubMed
Chimbira 1980
-
- Chimbira TH, Anderson ABM, Turnbull AC. Relation between measured menstrual blood loss and patient's subjective assessment of loss, duration of bleeding, number of sanitary towels used, uterine weight and endometrial surface area. British Journal of Obstetrics and Gynaecology 1980;87(7):603-9. - PubMed
Clegg 2007
-
- Clegg JP, Guest JF, Hurskainen R. Cost-utility of levonorgestrel intrauterine system compared with hysterectomy and second generation endometrial ablative techniques in managing patients with menorrhagia in the UK. Current Medical Research and Opinions 2007;23(7):1637-48. - PubMed
Cole 1971
-
- Cole SK, Billewicz WZ, Thomson AM. Sources of variation in menstrual blood loss. Journal of Obstetrics and Gynaecology of the British Commonwealth 1971;78(10):933-9. - PubMed
Cooper 2011
-
- Cooper K, Lee AJ, Chien P, Raja EA, Timmaraju V, Bhattacharya S. Outcomes following hysterectomy or endometrial ablation for heavy menstrual bleeding: retrospective analysis of hospital episode statistics in Scotland. British Journal of Obstetrics and Gynaecology 2011;118(10):1171-9. [DOI: 10.1111/j.1471-0528.2011.03011.x] - DOI - PubMed
Farquhar 2002
-
- Farquhar C, Steiner C. Hysterectomy rates in the United States 1990-1997. Obstetrics and Gynecology 2002;99(2):229-34. - PubMed
Fergusson 2013
Fraser 2009
-
- Fraser IS, Langham S, Uhl-Hochgraeber K. Health-related quality of life and economic burden of abnormal uterine bleeding. Expert Review of Obstetrics and Gynecology 2009;4(2):179-89.
Ganz 2013
-
- Ganz ML, Shah D, Gidwani R, Filonenko A, Su W, Pocoski J, et al. The cost effectiveness of the levonorgestrel-releasing intrauterine system for the treatment of idiopathic heavy menstrual bleeding in the United States. Value in Health 2013;16(2):325-33. - PubMed
Goldstuck 2017
GRADEpro GDT [Computer program]
-
- GRADEpro Guideline Development Tool. GRADEpro, Version accessed 4 July 2019. Hamilton (ON): McMaster University (developed by Evidence Prime), 2015.Available at gradepro.org. [ gradepro.org.]
Hallberg 1966
-
- Hallberg L, Hogahl AM, Nilsson L, Rybo G. Menstrual blood loss - a population study. Acta Obstetricia et Gynecologica Scandinavica 1966;45(3):320-51. - PubMed
Heikinheimo 2017
Higgins 2011
-
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0 [updated March 2011] edition. The Cochrane Collaboration, 2011.
Higham 1990
-
- Higham JM, O'Brien PMS, Shaw RW. Assessment of menstrual blood using a pictorial chart. British Journal of Obstetrics and Gynaecology 1990;97(8):734-9. - PubMed
Kaunitz 2009
-
- Kaunitz AM, Meredith S, Inki P, Kubba A, Sanchez-Ramos L. Levonorgestrel-releasing intrauterine system and endometrial ablation in heavy menstrual bleeding: a systematic review and meta-analysis. Obstetrics and Gynecology 2009;113(5):1104-16. - PubMed
Kaunitz 2012
-
- Kaunitz AM, Inki P. The levonorgestrel-releasing intrauterine system in heavy menstrual bleeding. Drugs 2012;72(2):193-215. - PubMed
Liu 2007
-
- Liu Z, Doan QV, Blumenthal P, Dubois RW. A systematic review evaluating health-related quality of life, work impairment, and health-care costs and utilisation in abnormal uterine bleeding. Value Health 2007;10(3):183-94. - PubMed
Matteson 2013
McNicholas 2015
Middleton 2010
-
- Middleton LJ, Champaneria R, Daniels JP, Bhattacharya S, Cooper KG, Hilken NH, et al. Hysterectomy, endometrial destruction and levonorgestrel releasing intrauterine system (Mirena) for heavy menstrual bleeding: systematic review and meta-analysis of data from individual patients. BMJ 2010;341:c3929. - PMC - PubMed
Moragianni 2007
-
- Moragianni VA, Somkuti SG. Profound hypothyroidism-induced acute menorrhagia resulting in life-threatening anemia. Obstetrics and Gynecology 2007;110(2 Pt 2):515-7. - PubMed
Munro 2012
NICE 2018
-
- National Institute for Health and Clinical Excellence. Heavy menstrual bleeding. pubmed.ncbi.nlm.nih.gov/21938862.
Nilsson 1978
-
- Nilsson CG, Luukkainen T, Arko H. Endometrial morphology of women using a d-norgestrel-releasing intrauterine device. Fertility and Sterility 1978;29(4):397-401. - PubMed
Reid 2005b
Roberts 2011
Rowe 2016
-
- Rowe P, Farley T, Peregoudov A, Piaggio G, Boccard S, Landoulsi S, et al. Safety and efficacy in parous women of a 52-mg levonorgestrel-medicated intrauterine device: a 7-year randomized comparative study with the TCu380A. Contraception 2016;93(6):498-506. [DOI: 10.1016/j.contraception.2016.02.024] - DOI - PMC - PubMed
Sergison 2019
Shapley 2004
Somboonporn 2011
-
- Somboonporn W, Panna S, Temtanakitpaisan T, Kaewrudee S, Soontrapa S. Effects of the levonorgestrel-releasing intrauterine system plus estrogen therapy in perimenopausal and postmenopausal women: systematic review and meta-analysis. Menopause 2011;18(10):1060-6. - PubMed
Suvisaari 1996
-
- Suvisaari J, Lahteenmaki P. Detailed analysis of menstrual bleeding patterns after postmenstrual and postabortal insertion of a copper IUD or a levonorgestrel-releasing intrauterine system. Contraception 1996;54(4):201-8. - PubMed
Wright 2013
Xu 2014
-
- Xu L, Lee BS, Asif S, Kraemer P, Inki P. Satisfaction and health-related quality of life in women with heavy menstrual bleeding; results from a non interventional trial of the levonorgestrel-releasing intrauterine system or conventional medical therapy. International Journal of Women's Health 2014;6:547-54. - PMC - PubMed
You 2006
-
- You JH, Sahota DS, MoYuen P. A cost utility analysis of hysterectomy, endometriation resection and ablation and medical therapy for menorrhagia. Human Reproduction 2006;21(7):1878-83. - PubMed
References to other published versions of this review
Lethaby 1999
Lethaby 2005
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

