Engineered Cell-Membrane-Coated Nanoparticles Directly Present Tumor Antigens to Promote Anticancer Immunity
- PMID: 32538494
- PMCID: PMC7669572
- DOI: 10.1002/adma.202001808
Engineered Cell-Membrane-Coated Nanoparticles Directly Present Tumor Antigens to Promote Anticancer Immunity
Abstract
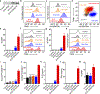
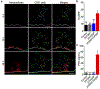
The recent success of immunotherapies has highlighted the power of leveraging the immune system in the fight against cancer. In order for most immune-based therapies to succeed, T cell subsets with the correct tumor-targeting specificities must be mobilized. When such specificities are lacking, providing the immune system with tumor antigen material for processing and presentation is a common strategy for stimulating antigen-specific T cell populations. While straightforward in principle, experience has shown that manipulation of the antigen presentation process can be incredibly complex, necessitating sophisticated strategies that are difficult to translate. Herein, the design of a biomimetic nanoparticle platform is reported that can be used to directly stimulate T cells without the need for professional antigen-presenting cells. The nanoparticles are fabricated using a cell membrane coating derived from cancer cells engineered to express a co-stimulatory marker. Combined with the peptide epitopes naturally presented on the membrane surface, the final formulation contains the necessary signals to promote tumor antigen-specific immune responses, priming T cells that can be used to control tumor growth. The reported approach represents an emerging strategy that can be used to develop multiantigenic, personalized cancer immunotherapies.
Keywords: anticancer vaccines; artificial antigen presentation; cell-membrane-coated nanoparticles; genetic engineering; immunotherapy.
© 2020 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
Conflict of Interest
The authors declare no conflict of interest.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

