Distinct Morphological Fates of Uropathogenic Escherichia coli Intracellular Bacterial Communities: Dependency on Urine Composition and pH
- PMID: 32540870
- PMCID: PMC7440767
- DOI: 10.1128/IAI.00884-19
Distinct Morphological Fates of Uropathogenic Escherichia coli Intracellular Bacterial Communities: Dependency on Urine Composition and pH
Abstract
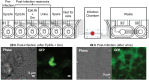
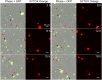
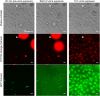
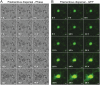
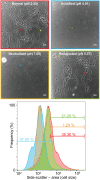
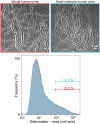
Uropathogenic Escherichia coli (UPEC) is the leading cause of urinary tract infections. These bacteria undertake a multistage infection cycle involving invasion of and proliferation within urinary tract epithelial cells, leading to the rupture of the host cell and dispersal of the bacteria, some of which have a highly filamentous morphology. Here, we established a microfluidics-based model of UPEC infection of immortalized human bladder epithelial cells that recapitulates the main stages of bacterial morphological changes during the acute infection cycle in vivo and allows the development and fate of individual cells to be monitored in real time by fluorescence microscopy. The UPEC-infected bladder cells remained alive and mobile in nonconfluent monolayers during the development of intracellular bacterial communities (IBCs). Switching from a flow of growth medium to human urine resulted in immobilization of both uninfected and infected bladder cells. Some IBCs continued to develop and then released many highly filamentous bacteria via an extrusion-like process, whereas other IBCs showed strong UPEC proliferation, and yet no filamentation was detected. The filamentation response was dependent on the weak acidity of human urine and required component(s) in a low molecular-mass (<3,000 Da) fraction from a mildly dehydrated donor. The developmental fate for bacteria therefore appears to be controlled by multiple factors that act at the level of the whole IBC, suggesting that variable local environments or stochastic differentiation pathways influence IBC developmental fates during infection.
Keywords: UPEC; infection model; microfluidics; morphological differentiation; stress response; urinary tract infection; urine.
Copyright © 2020 American Society for Microbiology.
Figures


References
-
- Phan MD, Peters KM, Sarkar S, Lukowski SW, Allsopp LP, Gomes Moriel D, Achard ME, Totsika M, Marshall VM, Upton M, Beatson SA, Schembri MA. 2013. The serum resistome of a globally disseminated multidrug resistant uropathogenic Escherichia coli clone. PLoS Genet 9:e1003834. doi:10.1371/journal.pgen.1003834. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

