Efficacy and Safety of Botulinum Toxin Type A on Persistent Myofascial Pain: A Randomized Clinical Trial
- PMID: 32549196
- PMCID: PMC7354430
- DOI: 10.3390/toxins12060395
Efficacy and Safety of Botulinum Toxin Type A on Persistent Myofascial Pain: A Randomized Clinical Trial
Abstract
This study assessed the safety and efficacy of three different doses of BoNT-A for persistent myofascial pain (MFP). One hundred female subjects were randomly assigned into five groups (n = 20): oral appliance (OA), saline solution (SS) and three BoNT-A groups with different doses. Pain intensity and pressure pain threshold were evaluated up to 24 weeks after treatment. Adverse effects related to muscle contraction, masticatory performance, muscle thickness and mandibular bone volume were also assessed. Changes over time were compared within and between groups. The "nparLD" package and Wilcoxon signed-rank test were used to analyze the data. BoNT-A reduced pain intensity (p < 0.0001) and increased pressure pain threshold (p < 0.0001) for up to 24 weeks compared to the placebo. No differences were found between BoNT-A and OA at the last follow-up. A transient decline in masticatory performance (p < 0.05) and muscle contraction (p < 0.0001), and a decrease in muscle thickness (p < 0.05) and coronoid and condylar process bone volume (p < 0.05) were found as dose-related adverse effects of BoNT-A. Regardless of the dose, BoNT-A was as effective as OA on MFP. Notwithstanding, due to BoNT-A dose-related adverse effects, we suggest the use of low doses of BoNT-A in MFP patients that do not benefit from conservative treatments.
Keywords: bone loss; botulinum toxin type A; chronic pain; myofascial pain; temporomandibular disorders.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
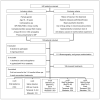


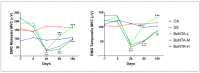
References
-
- Greene C.S. The etiology of temporomandibular disorders: Implications for treatment. J. Orofac. Pain. 2001;15:93–105. discussion 106–16. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

