Innervation: the missing link for biofabricated tissues and organs
- PMID: 32550009
- PMCID: PMC7275031
- DOI: 10.1038/s41536-020-0096-1
Innervation: the missing link for biofabricated tissues and organs
Abstract
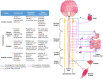
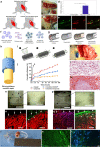
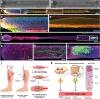
Innervation plays a pivotal role as a driver of tissue and organ development as well as a means for their functional control and modulation. Therefore, innervation should be carefully considered throughout the process of biofabrication of engineered tissues and organs. Unfortunately, innervation has generally been overlooked in most non-neural tissue engineering applications, in part due to the intrinsic complexity of building organs containing heterogeneous native cell types and structures. To achieve proper innervation of engineered tissues and organs, specific host axon populations typically need to be precisely driven to appropriate location(s) within the construct, often over long distances. As such, neural tissue engineering and/or axon guidance strategies should be a necessary adjunct to most organogenesis endeavors across multiple tissue and organ systems. To address this challenge, our team is actively building axon-based "living scaffolds" that may physically wire in during organ development in bioreactors and/or serve as a substrate to effectively drive targeted long-distance growth and integration of host axons after implantation. This article reviews the neuroanatomy and the role of innervation in the functional regulation of cardiac, skeletal, and smooth muscle tissue and highlights potential strategies to promote innervation of biofabricated engineered muscles, as well as the use of "living scaffolds" in this endeavor for both in vitro and in vivo applications. We assert that innervation should be included as a necessary component for tissue and organ biofabrication, and that strategies to orchestrate host axonal integration are advantageous to ensure proper function, tolerance, assimilation, and bio-regulation with the recipient post-implant.
Keywords: Regenerative medicine; Tissue engineering.
© The Author(s) 2020.
Conflict of interest statement
Competing interestsD.K.C and H.C.L. are co-founders of Axonova Medical, LLC, and D.K.C. is a co-founder of INNERVACE, Inc., which are University of Pennsylvania spin-out companies focused on translation of advanced regenerative therapies to treat nervous system disorders. Multiple patents relate to the composition, methods, and use of the constructs described in the paper, including U.S. Patent 9,895,399 (D.K.C.), U.S. Patent App. 15/032,677 (D.K.C.), U.S Provisional Patent App. 62/322,434 (D.K.C., H.I.C., M.D.S.), U.S. Provisional Patent 62/569,255 (D.K.C), and U.S. Provisional Patent App. 62/758,203 (D.K.C, S.D., W.G.V.). No other author declares competing interests.
Figures

References
-
- Mai, J. K. & Paxinos, G. The Human Nervous System (Academic Press, 2011).
-
- Cardinali, D. P. Autonomic Nervous System: Basic and Clinical Aspects (Springer, 2017).
-
- Mulroney, S. & Myers, A. Netter’s Essential Physiology E-Book (Elsevier Health Sciences, 2015).
-
- Furness JB, Callaghan BP, Rivera LR, Cho H-J. The enteric nervous system and gastrointestinal innervation: integrated local and central control. Adv. Exp. Med. Biol. 2014;817:39–71. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

