Targeting of Formyl Peptide Receptor 2 for in vivo imaging of acute vascular inflammation
- PMID: 32550892
- PMCID: PMC7295040
- DOI: 10.7150/thno.44226
Targeting of Formyl Peptide Receptor 2 for in vivo imaging of acute vascular inflammation
Abstract
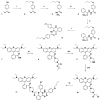
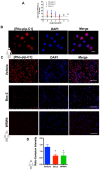
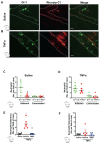
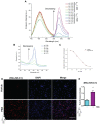
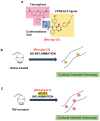
Inflammatory conditions are associated with a variety of diseases and can significantly contribute to their pathophysiology. Neutrophils are recognised as key players in driving vascular inflammation and promoting inflammation resolution. As a result, neutrophils, and specifically their surface formyl peptide receptors (FPRs), are attractive targets for non-invasive visualization of inflammatory disease states and studying mechanistic details of the process. Methods: A small-molecule Formyl Peptide Receptor 2 (FPR2/ALX)-targeted compound was combined with two rhodamine-derived fluorescent tags to form firstly, a targeted probe (Rho-pip-C1) and secondly a targeted, pH-responsive probe (Rho-NH-C1) for in vivo applications. We tested internalization, toxicity and functional interactions with neutrophils in vitro for both compounds, as well as the fluorescence switching response of Rho-NH-C1 to neutrophil activation. Finally, in vivo imaging (fluorescent intravital microscopy [IVM]) and therapeutic efficacy studies were performed in an inflammatory mouse model. Results: In vitro studies showed that the compounds bound to human neutrophils via FPR2/ALX without causing internalization at relevant concentrations. Additionally, the compounds did not cause toxicity or affect neutrophil functional responses (e.g. chemotaxis or transmigration). In vivo studies using IVM showed Rho-pip-C1 bound to activated neutrophils in a model of vascular inflammation. The pH-sensitive ("switchable") version termed Rho-NH-C1 validated these findings, showing fluorescent activity only in inflammatory conditions. Conclusions: These results indicate a viable design of fluorescent probes that have the ability to detect inflammatory events by targeting activated neutrophils.
Keywords: Inflammation; formyl peptide receptors; intravital microscopy; neutrophils; small-molecule imaging probes.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

