E-cigarette-induced pulmonary inflammation and dysregulated repair are mediated by nAChR α7 receptor: role of nAChR α7 in SARS-CoV-2 Covid-19 ACE2 receptor regulation
- PMID: 32552811
- PMCID: PMC7301079
- DOI: 10.1186/s12931-020-01396-y
E-cigarette-induced pulmonary inflammation and dysregulated repair are mediated by nAChR α7 receptor: role of nAChR α7 in SARS-CoV-2 Covid-19 ACE2 receptor regulation
Abstract
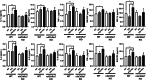
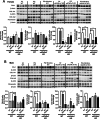
Electronic cigarette (e-cig) vaping is increasing rapidly in the United States, as e-cigs are considered less harmful than combustible cigarettes. However, limited research has been conducted to understand the possible mechanisms that mediate toxicity and pulmonary health effects of e-cigs. We hypothesized that sub-chronic e-cig exposure induces inflammatory response and dysregulated repair/extracellular matrix (ECM) remodeling, which occur through the α7 nicotinic acetylcholine receptor (nAChRα7). Adult wild-type (WT), nAChRα7 knockout (KO), and lung epithelial cell-specific KO (nAChRα7 CreCC10) mice were exposed to e-cig aerosol containing propylene glycol (PG) with or without nicotine. Bronchoalveolar lavage fluids (BALF) and lung tissues were collected to determine e-cig induced inflammatory response and ECM remodeling, respectively. Sub-chronic e-cig exposure with nicotine increased inflammatory cellular influx of macrophages and T-lymphocytes including increased pro-inflammatory cytokines in BALF and increased SARS-Cov-2 Covid-19 ACE2 receptor, whereas nAChRα7 KO mice show reduced inflammatory responses associated with decreased ACE2 receptor. Interestingly, matrix metalloproteinases (MMPs), such as MMP2, MMP8 and MMP9, were altered both at the protein and mRNA transcript levels in female and male KO mice, but WT mice exposed to PG alone showed a sex-dependent phenotype. Moreover, MMP12 was increased significantly in male mice exposed to PG with or without nicotine in a nAChRα7-dependent manner. Additionally, sub-chronic e-cig exposure with or without nicotine altered the abundance of ECM proteins, such as collagen and fibronectin, significantly in a sex-dependent manner, but without the direct role of nAChRα7 gene. Overall, sub-chronic e-cig exposure with or without nicotine affected lung inflammation and repair responses/ECM remodeling, which were mediated by nAChRα7 in a sex-dependent manner.
Keywords: Dysregulated repair; E-cig exposure; Extracellular matrix; Inflammation; nAChRα7.
Conflict of interest statement
The authors have declared that no competing interest.
Figures





Update of
-
E-cigarette-Induced Pulmonary Inflammation and Dysregulated Repair are Mediated by nAChR α7 Receptor: Role of nAChR α7 in ACE2 Covid-19 receptor regulation.Res Sq [Preprint]. 2020 May 18:rs.2.23829. doi: 10.21203/rs.2.23829/v2. Res Sq. 2020. Update in: Respir Res. 2020 Jun 18;21(1):154. doi: 10.1186/s12931-020-01396-y. PMID: 32702718 Free PMC article. Updated. Preprint.
References
-
- Madison MC, Landers CT, Gu BH, Chang CY, Tung HY, You R, Hong MJ, Baghaei N, Song LZ, Porter P, Putluri N, Salas R, Gilbert BE, Levental I, Campen MJ, Corry DB, and Kheradmand F. Electronic cigarettes disrupt lung lipid homeostasis and innate immunity independent of nicotine. J Clin Invest. 2019;129:4290–304. - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

