Meissner corpuscles and their spatially intermingled afferents underlie gentle touch perception
- PMID: 32554568
- PMCID: PMC7354383
- DOI: 10.1126/science.abb2751
Meissner corpuscles and their spatially intermingled afferents underlie gentle touch perception
Abstract
Meissner corpuscles are mechanosensory end organs that densely occupy mammalian glabrous skin. We generated mice that selectively lacked Meissner corpuscles and found them to be deficient in both perceiving the gentlest detectable forces acting on glabrous skin and fine sensorimotor control. We found that Meissner corpuscles are innervated by two mechanoreceptor subtypes that exhibit distinct responses to tactile stimuli. The anatomical receptive fields of these two mechanoreceptor subtypes homotypically tile glabrous skin in a manner that is offset with respect to one another. Electron microscopic analysis of the two Meissner afferents within the corpuscle supports a model in which the extent of lamellar cell wrappings of mechanoreceptor endings determines their force sensitivity thresholds and kinetic properties.
Copyright © 2020 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works.
Conflict of interest statement
Competing Interests
The authors have no competing interests.
Figures

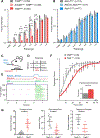
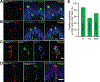



Comment in
-
Getting a grip on touch receptors.Science. 2020 Jun 19;368(6497):1311-1312. doi: 10.1126/science.abc7610. Science. 2020. PMID: 32554582 No abstract available.
References
-
- Wagner R, Meissner G, Über das Vorhandensein bisher unbekannter eigenthümlicher Tastkörperchen (Corpuscula tactus) in den Gefühlswarzchen der menschlichen Haut and über die Endausbreitung sensitiver Nerven. Nachricten von der Georg-August-Universität und der Königl Gesellschaft der Wissenschaften zu Göttingen 2, 17–30 (1852).
-
- González-Martínez T et al. , Absence of Meissner corpuscles in the digital pads of mice lacking functional TrkB. Brain Res 1002, 120–128 (2004). - PubMed
-
- González-Martínez T et al. , BDNF, but not NT-4, is necessary for normal development of Meissner corpuscles. Neurosci Lett 377, 12–15 (2005). - PubMed
-
- Perez-Pinera P et al. , Characterization of sensory deficits in TrkB knockout mice. Neurosci Lett 433, 43–47 (2008). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

