Immunomodulation to enhance the efficacy of an HPV therapeutic vaccine
- PMID: 32554612
- PMCID: PMC7304848
- DOI: 10.1136/jitc-2020-000612
Immunomodulation to enhance the efficacy of an HPV therapeutic vaccine
Abstract
Background: While prophylactic human papillomavirus (HPV) vaccines will certainly reduce the incidence of HPV-associated cancers, these malignancies remain a major health issue. PDS0101 is a liposomal-based HPV therapeutic vaccine consisting of the immune activating cationic lipid R-DOTAP and HLA-unrestricted HPV16 peptides that has shown in vivo CD8+ T cell induction and safety in a phase I study. In this report, we have employed the PDS0101 vaccine with two immune modulators previously characterized in preclinical studies and which are currently in phase II clinical trials. Bintrafusp alfa (M7824) is a first-in-class bifunctional fusion protein composed of the extracellular domains of the transforming growth factor-β receptor type II (TGFβRII) fused to a human IgG1 monoclonal antibody blocking programmed cell death protein-1 ligand (PDL1), designed both as a checkpoint inhibitor and to bring the TGFβRII 'trap' to the tumor microenvironment (TME). NHS-interleukin-12 (NHS-IL12) is a tumor targeting immunocytokine designed to bring IL-12 to the TME and thus enhance the inflammatory Th1 response.
Methods: We employed TC-1 carcinoma (expressing HPV16 E6 and E7 and devoid of PDL1 expression) in a syngeneic mouse model in monotherapy and combination therapy studies to analyze antitumor effects and changes in immune cell types in the spleen and the TME.
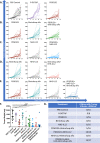
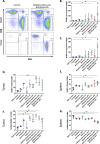
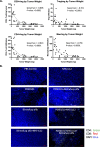
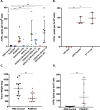
Results: As a monotherapy, the PDS0101 vaccine generated HPV-specific T cells and antitumor activity in mice bearing HPV-expressing mEER oropharyngeal and TC-1 lung carcinomas. When used as a monotherapy in the TC-1 model, NHS-IL12 elicited antitumor effects as well as an increase in CD8+ T cells in the TME. When used as a monotherapy, bintrafusp alfa did not elicit antitumor effects or any increase in T cells in the TME. When all three agents were used in combination, maximum antitumor effects were observed, which correlated with increases in T cells and T-cell clonality in the TME.
Conclusion: These studies provide the rationale for the potential clinical use of combinations of agents that can (1) induce tumor-associated T-cell responses, (2) potentiate immune responses in the TME and (3) reduce immunosuppressive entities in the TME.
Keywords: genital Neoplasms, female; head and neck neoplasms; immunotherapy; therapies, investigational; vaccination.
© Author(s) (or their employer(s)) 2020. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declare.
Figures

Similar articles
-
Development of DNA Vaccine Targeting E6 and E7 Proteins of Human Papillomavirus 16 (HPV16) and HPV18 for Immunotherapy in Combination with Recombinant Vaccinia Boost and PD-1 Antibody.mBio. 2021 Jan 19;12(1):e03224-20. doi: 10.1128/mBio.03224-20. mBio. 2021. PMID: 33468698 Free PMC article.
-
Induction of systemic immune responses and reversion of immunosuppression in the tumor microenvironment by a therapeutic vaccine for cervical cancer.Cancer Immunol Immunother. 2020 Dec;69(12):2651-2664. doi: 10.1007/s00262-020-02651-3. Epub 2020 Jul 1. Cancer Immunol Immunother. 2020. PMID: 32607768 Free PMC article.
-
Mucosal HPV E6/E7 Peptide Vaccination in Combination with Immune Checkpoint Modulation Induces Regression of HPV+ Oral Cancers.Cancer Res. 2018 Sep 15;78(18):5327-5339. doi: 10.1158/0008-5472.CAN-18-0892. Epub 2018 Jul 27. Cancer Res. 2018. PMID: 30054333 Free PMC article.
-
Preclinical and clinical studies of bintrafusp alfa, a novel bifunctional anti-PD-L1/TGFβRII agent: Current status.Exp Biol Med (Maywood). 2022 Jul;247(13):1124-1134. doi: 10.1177/15353702221089910. Epub 2022 Apr 27. Exp Biol Med (Maywood). 2022. PMID: 35473390 Free PMC article. Review.
-
Comparison of preclinical efficacy of immunotherapies against HPV-induced cancers.Expert Rev Vaccines. 2024 Jan-Dec;23(1):674-687. doi: 10.1080/14760584.2024.2374287. Epub 2024 Jul 8. Expert Rev Vaccines. 2024. PMID: 38978164 Review.
Cited by
-
Computationally Optimized Hemagglutinin Proteins Adjuvanted with Infectimune® Generate Broadly Protective Antibody Responses in Mice and Ferrets.Vaccines (Basel). 2024 Dec 2;12(12):1364. doi: 10.3390/vaccines12121364. Vaccines (Basel). 2024. PMID: 39772026 Free PMC article.
-
Nanoparticle-based applications for cervical cancer treatment in drug delivery, gene editing, and therapeutic cancer vaccines.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2021 Sep;13(5):e1718. doi: 10.1002/wnan.1718. Epub 2021 May 4. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2021. PMID: 33942532 Free PMC article. Review.
-
R-DOTAP Cationic Lipid Nanoparticles Outperform Squalene-Based Adjuvant Systems in Elicitation of CD4 T Cells after Recombinant Influenza Hemagglutinin Vaccination.Viruses. 2023 Feb 15;15(2):538. doi: 10.3390/v15020538. Viruses. 2023. PMID: 36851752 Free PMC article.
-
A Newcastle Disease Virus (NDV) Expressing a Membrane-Anchored Spike as a Cost-Effective Inactivated SARS-CoV-2 Vaccine.Vaccines (Basel). 2020 Dec 17;8(4):771. doi: 10.3390/vaccines8040771. Vaccines (Basel). 2020. PMID: 33348607 Free PMC article.
-
Precision therapeutic targets for HPV-positive cancers: an overview and new insights.Infect Agent Cancer. 2025 Mar 11;20(1):17. doi: 10.1186/s13027-025-00641-7. Infect Agent Cancer. 2025. PMID: 40069817 Free PMC article. Review.
References
-
- Centers for Disease Control and Prevention. Cancers Associated with Human Papillomavirus, United States—2012–2016 U.S. Cancer Statistics Data Brief. Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services, 2019.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
