KEYNOTE-013 4-year follow-up of pembrolizumab in classical Hodgkin lymphoma after brentuximab vedotin failure
- PMID: 32556281
- PMCID: PMC7322954
- DOI: 10.1182/bloodadvances.2019001367
KEYNOTE-013 4-year follow-up of pembrolizumab in classical Hodgkin lymphoma after brentuximab vedotin failure
Abstract
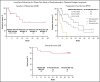
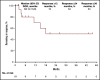
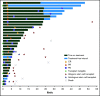
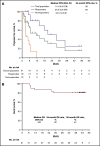
The KEYNOTE-013 study was conducted to evaluate pembrolizumab monotherapy in hematologic malignancies; classical Hodgkin lymphoma (cHL) was an independent expansion cohort. We present long-term results based on >4 years of median follow-up for the cHL cohort. The trial enrolled cHL patients who experienced relapse after, were ineligible for, or declined autologous stem cell transplantation and experienced progression with or did not respond to brentuximab vedotin. Patients received IV pembrolizumab 10 mg/kg every 2 weeks for up to 2 years or until confirmed progression or unacceptable toxicity. Primary end points were safety and complete response (CR) rate by central review. Enrolled patients (N = 31) had received a median of 5 therapies (range, 2 to 15). After a median follow-up of 52.8 months (range, 7.0 to 57.6 months), CR rate was 19%, and median duration of response (DOR) was not reached; 24-month and 36-month DOR rates were both 50% by the Kaplan-Meier method. Median overall survival was not reached; 36-month overall survival was 81%. Six patients (19%) experienced grade 3 treatment-related adverse events (AEs); there were no grade 4 or 5 treatment-related AEs. With long-term follow-up among a heavily pretreated cohort, pembrolizumab had a favorable safety profile; some patients maintained long-term response with pembrolizumab years after end of treatment. This trial was registered at www.clinicaltrials.gov as #NCT01953692.
© 2020 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: P.A. has received honoraria from Merck & Co., Inc., Bristol-Myers Squibb, Pfizer, Affimed, Adaptive, Infinity, ADC Therapeutics, Celgene, Morphosys, Daiichi Sankyo, Miltenyi, Tessa, GenMab, C4, and Enterome and has received research funding from Merck & Co., Inc., Bristol-Myers Squibb, Affimed, Adaptive, Roche, Tensha, Otsuka, Sigma Tau, Genentech, and IGM. J.K. has been a consultant for Merck & Co., Inc., and has received payment for lectures and speakers’ bureaus from Merck & Co., Inc. V.R. has been a consultant/advisor for Gilead, Infinity, Bristol-Myers Squibb, NanoString, and Incyte; has received research funding from ArgenX; has provided expert testimony for Servier; and has received travel, accommodations, and expenses from Roche and Bristol-Myers Squibb. P.L.Z. has served on speakers’ bureaus for Verastem, Celltrion, Gilead, Janssen-Cilag, Bristol-Myers Squibb, Servier, Sandoz, Merck & Co., Inc., Immune Design, Celgene, Portola, Roche, Eusapharma, Kyowa Kirin, and Sanofi; has served on advisory boards for Verastem, Celltrion, Gilead, Janssen-Cilag, Bristol-Myers Squibb, Servier, Merck & Co., Inc., Immune Design, Celgene, Portola, Roche, Eusapharma, Kyowa Kirin, and Sanofi; and has been a consultant for Verastem, Merck & Co., Inc., Eusapharma, and Sanofi. Y.Z. and A.N. are employees of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ. P.M. is an employee and stockholder of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, and has received travel expenses from Merck & Co., Inc. C.H.M. has held an employment/leadership position/advisory role for Celgene, Genentech, Merck & Co., Seattle Genetics Inc., participated in an advisory board for Molecular Templates, and has received research funding from Pharmacyclics, Genentech, Merck & Co., Inc., and Seattle Genetics Inc. J.-M.M. declares no competing financial interests.
Figures
References
-
- Elshenawy M, Shahzad Rauf M, Elhassan T, Maghfoor I, Akhtar S. Survival analysis of patients with Hodgkin lymphoma who failed high dose chemotherapy and autologous stem cell transplant. Ann Hematol. 2018;97(7):1229-1240. - PubMed

