Enteropathogenic Escherichia coli Infection Inhibits Intestinal Ascorbic Acid Uptake via Dysregulation of Its Transporter Expression
- PMID: 32556816
- PMCID: PMC7744340
- DOI: 10.1007/s10620-020-06389-x
Enteropathogenic Escherichia coli Infection Inhibits Intestinal Ascorbic Acid Uptake via Dysregulation of Its Transporter Expression
Abstract
Background: Enteropathogenic Escherichia coli (EPEC) infection causes prolonged, watery diarrhea leading to morbidity and mortality. Although EPEC infection impacts nutrient transporter function and expression in intestinal epithelial cells, the effects of EPEC infection on intestinal absorption of ascorbic acid (AA) have not yet been investigated.
Aims: To investigate the effect of EPEC infection on intestinal AA uptake process and expression of both AA transporters.
Methods: We used two experimental models: human-derived intestinal epithelial Caco-2 cells and mice. 14C-AA uptake assay, Western blot, RT-qPCR, and promoter assay were performed.
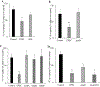
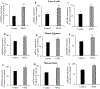
Results: EPEC (WT) as well as ΔespF and ΔespG/G2 mutant-infected Caco-2 cells showed markedly inhibited AA uptake, while other mutants (ΔescN, ΔespA, ΔespB, and ΔespD) did not affect AA uptake. Infection also reduced protein and mRNA expression levels for both hSVCT1 and hSVCT2. EPEC-infected mice showed marked inhibitory effect on AA uptake and decreased protein and mRNA expression levels for both mSVCT1 and mSVCT2 in jejunum and colon. MicroRNA regulators of SVCT1 and SVCT2 (miR103a, miR141, and miR200a) were upregulated significantly upon EPEC infection in both Caco-2 and mouse jejunum and colon. In addition, expression of the accessory protein glyoxalate reductase/hydroxypyruvate reductase (GRHPR), which regulates SVCT1 function, was markedly decreased by EPEC infection in both models.
Conclusions: These findings suggest that EPEC infection causes inhibition in AA uptake through a multifactorial dysregulation of SVCT1 and SVCT2 expression in intestinal epithelial cells.
Keywords: EPEC; GRHPR; SVCT1; SVCT2; Vitamin C; microRNA.
Conflict of interest statement
Figures


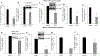
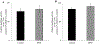
Comment in
-
I Can C Clearly Now: How EPEC Inhibits Gut Vitamin C Transport by Dysregulating SVCT.Dig Dis Sci. 2021 Jul;66(7):2140-2142. doi: 10.1007/s10620-020-06594-8. Epub 2020 Sep 12. Dig Dis Sci. 2021. PMID: 32918648 Free PMC article. No abstract available.
Similar articles
-
Enterotoxigenic Escherichia coli heat labile enterotoxin inhibits intestinal ascorbic acid uptake via a cAMP-dependent NF-κB-mediated pathway.Am J Physiol Gastrointest Liver Physiol. 2019 Jan 1;316(1):G55-G63. doi: 10.1152/ajpgi.00259.2018. Epub 2018 Oct 4. Am J Physiol Gastrointest Liver Physiol. 2019. PMID: 30285481 Free PMC article.
-
Modulation of function of sodium-dependent vitamin C transporter 1 (SVCT1) by Rab8a in intestinal epithelial cells: studies utilizing Caco-2 cells and Rab8a knockout mice.Dig Dis Sci. 2013 Mar;58(3):641-9. doi: 10.1007/s10620-012-2388-9. Epub 2012 Sep 27. Dig Dis Sci. 2013. PMID: 23014846 Free PMC article.
-
Enteropathogenic Escherichia coli inhibits intestinal vitamin B1 (thiamin) uptake: studies with human-derived intestinal epithelial Caco-2 cells.Am J Physiol Gastrointest Liver Physiol. 2009 Oct;297(4):G825-33. doi: 10.1152/ajpgi.00250.2009. Epub 2009 Jul 23. Am J Physiol Gastrointest Liver Physiol. 2009. PMID: 19628653 Free PMC article.
-
The sodium-dependent ascorbic acid transporter family SLC23.Mol Aspects Med. 2013 Apr-Jun;34(2-3):436-54. doi: 10.1016/j.mam.2012.12.002. Mol Aspects Med. 2013. PMID: 23506882 Review.
-
[Molecular pathogenesis, epidemiology and diagnosis of enteropathogenic Escherichia coli].Salud Publica Mex. 2007 Sep-Oct;49(5):376-86. doi: 10.1590/s0036-36342007000500008. Salud Publica Mex. 2007. PMID: 17952245 Review. Spanish.
Cited by
-
Effect of Lipopolysaccharide and TNFα on Neuronal Ascorbic Acid Uptake.Mediators Inflamm. 2021 Jul 3;2021:4157132. doi: 10.1155/2021/4157132. eCollection 2021. Mediators Inflamm. 2021. PMID: 34285658 Free PMC article.
-
Ascorbate deficiency increases progression of shigellosis in guinea pigs and mice infection models.Gut Microbes. 2023 Dec;15(2):2271597. doi: 10.1080/19490976.2023.2271597. Epub 2023 Oct 24. Gut Microbes. 2023. PMID: 37876025 Free PMC article.
-
The Variable Nature of Vitamin C-Does It Help When Dealing with Coronavirus?Antioxidants (Basel). 2022 Jun 24;11(7):1247. doi: 10.3390/antiox11071247. Antioxidants (Basel). 2022. PMID: 35883738 Free PMC article. Review.
-
Vitamins as regulators of calcium-containing kidney stones - new perspectives on the role of the gut microbiome.Nat Rev Urol. 2023 Oct;20(10):615-637. doi: 10.1038/s41585-023-00768-5. Epub 2023 May 9. Nat Rev Urol. 2023. PMID: 37161031 Free PMC article. Review.
-
Calsyntenin-3 interacts with the sodium-dependent vitamin C transporter-2 to regulate vitamin C uptake.Int J Biol Macromol. 2021 Dec 1;192:1178-1184. doi: 10.1016/j.ijbiomac.2021.10.058. Epub 2021 Oct 18. Int J Biol Macromol. 2021. PMID: 34673103 Free PMC article.
References
-
- Packer L, Fuchs J. Vitamin C in health and disease. Marcel Dekker Inc, New York, USA. 1997.
-
- Carr AC, Frei B. Toward a new recommended dietary allowance for Vitamin C based on antioxidant and health effects in humans. Am J Clin Nutr. 1999; 69:1086–1107. - PubMed
-
- Simon JA, Hudes ES. Serum ascorbic acid and gallbladder disease prevalence among US adults. Arch Intern Med. 2000; 160:931–936. - PubMed
-
- Harrison SA, Torgerson S, Hayashi P, et al. Vitamin E and vitamin C treatment improves fibrosis in patients with nonalcoholic steatohepatitis. Am J Gastroenterol. 2003; 98:2485–2490. - PubMed

