An astrocyte cell line that differentially propagates murine prions
- PMID: 32561641
- PMCID: PMC7450132
- DOI: 10.1074/jbc.RA120.012596
An astrocyte cell line that differentially propagates murine prions
Abstract
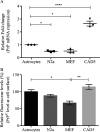
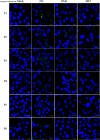
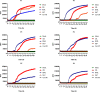
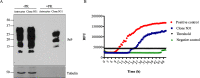
Prion diseases are fatal infectious neurodegenerative disorders in human and animals caused by misfolding of the cellular prion protein (PrPC) into the pathological isoform PrPSc Elucidating the molecular and cellular mechanisms underlying prion propagation may help to develop disease interventions. Cell culture systems for prion propagation have greatly advanced molecular insights into prion biology, but translation of in vitro to in vivo findings is often disappointing. A wider range of cell culture systems might help overcome these shortcomings. Here, we describe an immortalized mouse neuronal astrocyte cell line (C8D1A) that can be infected with murine prions. Both PrPC protein and mRNA levels in astrocytes were comparable with those in neuronal and non-neuronal cell lines permitting persistent prion infection. We challenged astrocytes with three mouse-adapted prion strains (22L, RML, and ME7) and cultured them for six passages. Immunoblotting results revealed that the astrocytes propagated 22L prions well over all six passages, whereas ME7 prions did not replicate, and RML prions replicated only very weakly after five passages. Immunofluorescence analysis indicated similar results for PrPSc Interestingly, when we used prion conversion activity as a readout in real-time quaking-induced conversion assays with RML-infected cell lysates, we observed a strong signal over all six passages, comparable with that for 22L-infected cells. These data indicate that the C8D1A cell line is permissive to prion infection. Moreover, the propagated prions differed in conversion and proteinase K-resistance levels in these astrocytes. We propose that the C8D1A cell line could be used to decipher prion strain biology.
Keywords: C8D1A; Creutzfeldt–Jakob disease; astrocyte; astrocytes; bovine spongiform encephalopathy; neurodegeneration; neurodegenerative disease; prion; prion disease; prion infection; prion strain; protein misfolding; scrapie.
© 2020 Tahir et al.
Conflict of interest statement
Conflict of interest—Conflict of interest statement: The authors declare that they have no conflicts of interest with the contents of this article.
Figures

References
-
- Raymond G. J., Hope J., Kocisko D. A., Priola S. A., Raymond L. D., Bossers A., Ironside J., Will R. G., Chen S. G., Petersen R. B., Gambetti P., Rubenstein R., Smits M. A., Lansbury P. T., and Caughey B. (1997) Molecular assessment of the potential transmissibilities of BSE and scrapie to humans. Nature 388, 285–288 10.1038/40876 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

