Causes and Consequences of Snake Venom Variation
- PMID: 32564899
- PMCID: PMC7116101
- DOI: 10.1016/j.tips.2020.05.006
Causes and Consequences of Snake Venom Variation
Abstract
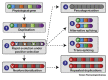
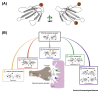
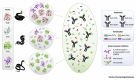
Snake venoms are mixtures of toxins that vary extensively between and within snake species. This variability has serious consequences for the management of the world's 1.8 million annual snakebite victims. Advances in 'omic' technologies have empowered toxinologists to comprehensively characterize snake venom compositions, unravel the molecular mechanisms that underpin venom variation, and elucidate the ensuing functional consequences. In this review, we describe how such mechanistic processes have resulted in suites of toxin isoforms that cause diverse pathologies in human snakebite victims and we detail how variation in venom composition can result in treatment failure. Finally, we outline current therapeutic approaches designed to circumvent venom variation and deliver next-generation treatments for the world's most lethal neglected tropical disease.
Keywords: antivenom; gene duplication; protein neofunctionalization; snakebite; toxins; venom evolution.
Copyright © 2020 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures
References
-
- Post Y, et al. Snake venom gland organoids. Cell. 2020;180:233–247. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

