Physiological Effect of Ghrelin on Body Systems
- PMID: 32565790
- PMCID: PMC7267865
- DOI: 10.1155/2020/1385138
Physiological Effect of Ghrelin on Body Systems
Abstract
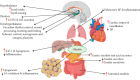
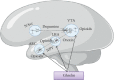
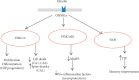
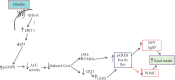
Ghrelin is a relatively novel multifaceted hormone that has been found to exert a plethora of physiological effects. In this review, we found/confirmed that ghrelin has effect on all body systems. It induces appetite; promotes the use of carbohydrates as a source of fuel while sparing fat; inhibits lipid oxidation and promotes lipogenesis; stimulates the gastric acid secretion and motility; improves cardiac performance; decreases blood pressure; and protects the kidneys, heart, and brain. Ghrelin is important for learning, memory, cognition, reward, sleep, taste sensation, olfaction, and sniffing. It has sympatholytic, analgesic, antimicrobial, antifibrotic, and osteogenic effects. Moreover, ghrelin makes the skeletal muscle more excitable and stimulates its regeneration following injury; delays puberty; promotes fetal lung development; decreases thyroid hormone and testosterone; stimulates release of growth hormone, prolactin, glucagon, adrenocorticotropic hormone, cortisol, vasopressin, and oxytocin; inhibits insulin release; and promotes wound healing. Ghrelin protects the body by different mechanisms including inhibition of unwanted inflammation and induction of autophagy. Having a clear understanding of the ghrelin effect in each system has therapeutic implications. Future studies are necessary to elucidate the molecular mechanisms of ghrelin actions as well as its application as a GHSR agonist to treat most common diseases in each system without any paradoxical outcomes on the other systems.
Copyright © 2020 Yonas Akalu et al.
Conflict of interest statement
None of the authors have conflicts of interest to declare.
Figures




References
-
- Paress P. S., McKee K. K., Liu K. K., et al. A receptor in pituitary and hypothalamus that functions in growth hormone release. Science. 2006;273(5277):974–977. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

