Growth Differentiation Factor 15 is a potential biomarker of therapeutic response for TK2 deficient myopathy
- PMID: 32572108
- PMCID: PMC7308386
- DOI: 10.1038/s41598-020-66940-8
Growth Differentiation Factor 15 is a potential biomarker of therapeutic response for TK2 deficient myopathy
Abstract
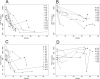
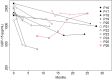
GDF-15 is a biomarker for mitochondrial diseases. We investigated the application of GDF-15 as biomarker of disease severity and response to deoxynucleoside treatment in patients with thymidine kinase 2 (TK2) deficiency and compared it to FGF-21. GDF-15 and FGF-21 were measured in serum from 24 patients with TK2 deficiency treated 1-49 months with oral deoxynucleosides. Patients were grouped according to age at treatment and biomarkers were analyzed at baseline and various time points after treatment initiation. GDF-15 was elevated on average 30-fold in children and 6-fold in adults before the start of treatment. There was a significant correlation between basal GDF-15 and severity based on pretreatment distance walked (6MWT) and weight (BMI). During treatment, GDF-15 significantly declined, and the decrease was accompanied by relevant clinical improvements. The decline was greater in the paediatric group, which included the most severe patients and showed the greatest clinical benefit, than in the adult patients. The decline of FGF-21 was less prominent and consistent. GDF-15 is a potential biomarker of severity and of therapeutic response for patients with TK2 deficiency. In addition, we show evidence of clinical benefit of deoxynucleoside treatment, especially when treatment is initiated at an early age.
Conflict of interest statement
MH, RM, Columbia University Medical Center (CUMC), and the Vall d’Hebron Research Institute (VHIR) have filed patent applications for deoxynucleoside and deoxynucleotide therapies for human mitochondrial DNA depletion and deletions syndrome including TK2 deficiency. RM, YC, CB, VHIR, and The Biomedical Network Research Centre on Rare Diseases (CIBERER) have filed patent applications covering potential use of deoxynucleoside treatment for POLG deficiency and other mtDNA replication defects in humans. CUMC, VHIR, and CIBERER have licensed pending patent applications related to these technologies to Modis Therapeutics, Inc. CUMC, VHIR, and CIBERER may be eligible to receive payments related to the development and commercialization of the technologies. Any potential licensing fees earned will be paid to CUMC, VHIR, and CIBERER and are shared with all inventors mentioned above through VHIR and CIBERER policies on distribution and objectivity in research. MH and RM serve as paid consultants to Modis Therapeutics, Inc. and RM has equity in this company. RM, YC, CB, VHIR, and The Biomedical Network Research Centre on Rare Diseases (CIBERER) have filed patent applications covering the potential use of deoxynucleoside treatment as a way to increase mtDNA copy number.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

