The Effect of Denosumab on Bone Mass in Super Elderly Patients
- PMID: 32572372
- PMCID: PMC7297623
- DOI: 10.11005/jbm.2020.27.2.119
The Effect of Denosumab on Bone Mass in Super Elderly Patients
Abstract
Background: Denosumab is a potent antiresorptive drug leading to significant reduction in the risk of vertebral and non-vertebral fractures in postmenopausal osteoporosis. The effect of denosumab in super-elderly patients lacks data to date and few literature has proven the efficacy to this specific group. The purpose of this study was to determine the effectiveness and safety of denosumab in the super-elderly.
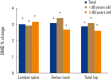
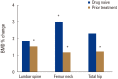
Methods: We retrospectively evaluated 60 patients older than 80 with osteoporosis treated with denosumab. Patients were treated with denosumab every 6 months for 12 months 2017 to 2020. The primary endpoint was defined by the changes in bone mineral density (BMD) of 3 measurement sites: the lumbar spine, femoral neck, and total hip. Changes in bone turnover markers, serum calcium, serum phosphate, and 25-hydroxy-vitamin D were also observed.
Results: All 60 patients were female, and the mean age was 83.9±3.1, from age 80 to 94. After 12 months of denosumab treatment, significant increases in BMD were observed; 3.02±2.74% for the lumbar spine (P=0.000), 3.10±6.90% for the femoral neck (P=0.005), and 2.89±5.80% for the total hip (P=0.002) The bone turnover marker C-terminal telopeptide of type I collagen and osteocalcin significantly declined after 12 months of treatment (-34.8±45.9%; P=0.002 and -35.5±38.9%; P=0.004 respectively). Symptomatic hypocalcemia and serious adverse drug reactions that required drug discontinuation were not observed during treatment.
Conclusions: Denosumab is thought to be an anti-osteoporotic medication that is sufficiently effective and safe even for the super-elderly.
Keywords: Aged; Bone density; Denosumab; Osteoporosis.
Copyright © 2020 The Korean Society for Bone and Mineral Research.
Conflict of interest statement
Conflict of interest: No potential conflict of interest relevant to this article was reported.
Figures
References
-
- United Nations, Department of Economic and Social Affairs, Population Division. World population prospects 2019: Highlights (ST/ESA/SER.A/423) 2019. [cited by 2020 Jan 3]. Available from: https://population.un.org/wpp/Publications/Files/WPP2019_Highlights.pdf.
-
- Ettinger MP. Aging bone and osteoporosis: strategies for preventing fractures in the elderly. Arch Intern Med. 2003;163:2237–2246. - PubMed
-
- Kanis JA, Johnell O, Oden A, et al. Ten year probabilities of osteoporotic fractures according to BMD and diagnostic thresholds. Osteoporos Int. 2001;12:989–995. - PubMed
-
- Ström O, Borgstrom F, Kanis JA, et al. Osteoporosis: burden, health care provision and opportunities in the EU: a report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA) Arch Osteoporos. 2011;6:59–155. - PubMed
-
- Wilkins CH, Birge SJ. Prevention of osteoporotic fractures in the elderly. Am J Med. 2005;118:1190–1195. - PubMed

