A new genetic strategy for targeting microglia in development and disease
- PMID: 32573436
- PMCID: PMC7375817
- DOI: 10.7554/eLife.54590
A new genetic strategy for targeting microglia in development and disease
Abstract
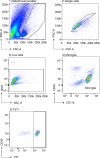
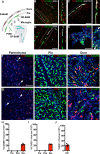
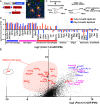
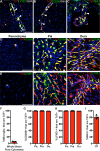
As the resident macrophages of the brain and spinal cord, microglia are crucial for the phagocytosis of infectious agents, apoptotic cells and synapses. During brain injury or infection, bone-marrow derived macrophages invade neural tissue, making it difficult to distinguish between invading macrophages and resident microglia. In addition to circulation-derived monocytes, other non-microglial central nervous system (CNS) macrophage subtypes include border-associated meningeal, perivascular and choroid plexus macrophages. Using immunofluorescent labeling, flow cytometry and Cre-dependent ribosomal immunoprecipitations, we describe P2ry12-CreER, a new tool for the genetic targeting of microglia. We use this new tool to track microglia during embryonic development and in the context of ischemic injury and neuroinflammation. Because of the specificity and robustness of microglial recombination with P2ry12-CreER, we believe that this new mouse line will be particularly useful for future studies of microglial function in development and disease.
Keywords: fate mapping; immunology; inflammation; microglia; mouse; multiple sclerosis; neuroinflammation; neuroscience; ribosomal profiling; stroke.
© 2020, McKinsey et al.
Conflict of interest statement
GM, CL, AK, AN, NS, AL, EC, FG, TA No competing interests declared
Figures










References
-
- Arnold TD, Lizama CO, Cautivo KM, Santander N, Lin L, Qiu H, Huang EJ, Liu C, Mukouyama YS, Reichardt LF, Zovein AC, Sheppard D. Impaired αvβ8 and tgfβ signaling lead to microglial dysmaturation and neuromotor dysfunction. Journal of Experimental Medicine. 2019;216:900–915. doi: 10.1084/jem.20181290. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

