Facile One-Step Electrodeposition Preparation of Cationic Pillar[6]arene-Modified Graphene Films on Glassy Carbon Electrodes for Enhanced Electrochemical Performance
- PMID: 32582632
- PMCID: PMC7287394
- DOI: 10.3389/fchem.2020.00430
Facile One-Step Electrodeposition Preparation of Cationic Pillar[6]arene-Modified Graphene Films on Glassy Carbon Electrodes for Enhanced Electrochemical Performance
Abstract
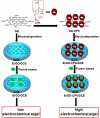
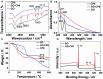
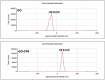
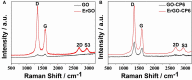
In the present work, we have developed a facile one-step route for preparing electrochemically reduced graphene oxide-cationic pillar[6]arene (ErGO-CP6) nanocomposite films on glassy carbon electrodes (GCEs) directly from graphene oxide-cationic pillar[6]arene (GO-CP6) colloidal solution by using a pulsed electrodeposition technique. The electrocatalytic activity of ErGO-CP6 was examined by studying the oxidations of five purine bases [adenine (A), guanine (G), xanthine (X), hypoxanthine (HX), and uric acid (UA)]. It enhanced the oxidation currents of A, G, X, HX, and UA when compared to unmodified ErGO films and bare GCE, which is considered to be the synergetic effects of the graphene (excellent electrical properties and large surface area) and CP6 molecules (high inclusion complexation and enrichment capability).
Keywords: electrochemical performance; electrodeposition; graphene films; host-guest inclusion; pillar[6]arene.
Copyright © 2020 Duan, Wang, Wang, Zhang and Lu.
Figures




Similar articles
-
Direct electrodeposition of cationic pillar[6]arene-modified graphene oxide composite films and their host-guest inclusions for enhanced electrochemical performance.RSC Adv. 2020 Jun 9;10(37):21954-21962. doi: 10.1039/d0ra03138k. eCollection 2020 Jun 8. RSC Adv. 2020. PMID: 35516626 Free PMC article.
-
Electrodeposited reduced graphene oxide incorporating polymerization of l-lysine on electrode surface and its application in simultaneous electrochemical determination of ascorbic acid, dopamine and uric acid.Mater Sci Eng C Mater Biol Appl. 2017 Jan 1;70(Pt 1):241-249. doi: 10.1016/j.msec.2016.08.078. Epub 2016 Aug 31. Mater Sci Eng C Mater Biol Appl. 2017. PMID: 27770887
-
Simultaneous determination of uric acid, xanthine, hypoxanthine and caffeine in human blood serum and urine samples using electrochemically reduced graphene oxide modified electrode.Anal Chim Acta. 2013 Apr 10;771:14-20. doi: 10.1016/j.aca.2013.02.017. Epub 2013 Feb 19. Anal Chim Acta. 2013. PMID: 23522107
-
Electrochemical behavior and voltammetric determination of acetaminophen based on glassy carbon electrodes modified with poly(4-aminobenzoic acid)/electrochemically reduced graphene oxide composite films.Mater Sci Eng C Mater Biol Appl. 2014 Dec;45:21-8. doi: 10.1016/j.msec.2014.08.067. Epub 2014 Sep 6. Mater Sci Eng C Mater Biol Appl. 2014. PMID: 25491797
-
Novel Electrochemical Sensors Based on Cuprous Oxide-Electrochemically Reduced Graphene Oxide Nanocomposites Modified Electrode toward Sensitive Detection of Sunset Yellow.Molecules. 2018 Aug 24;23(9):2130. doi: 10.3390/molecules23092130. Molecules. 2018. PMID: 30149513 Free PMC article.
Cited by
-
A Sensitive and Selective Electrochemical Aptasensor for Carbendazim Detection.Biosensors (Basel). 2025 Jan 3;15(1):15. doi: 10.3390/bios15010015. Biosensors (Basel). 2025. PMID: 39852066 Free PMC article.
-
Correction: Direct electrodeposition of cationic pillar[6]arene-modified graphene oxide composite films and their host-guest inclusions for enhanced electrochemical performance.RSC Adv. 2020 Dec 18;10(73):44834. doi: 10.1039/d0ra90132f. eCollection 2020 Dec 17. RSC Adv. 2020. PMID: 35532458 Free PMC article.
-
Enhancing the Stability and Photothermal Conversion Efficiency of ICG by Pillar[5]arene-Based Host-Guest Interaction.Front Chem. 2021 Oct 29;9:775436. doi: 10.3389/fchem.2021.775436. eCollection 2021. Front Chem. 2021. PMID: 34778221 Free PMC article.
References
-
- Aparna T. K., Sivasubramanian R., Dar M. A. (2018). One-pot synthesis of Au-Cu2O/rGO nanocomposite based electrochemical sensor for selective and simultaneous detection of dopamine and uric acid. J. Alloys Compd. 741, 1130–1141. 10.1016/j.jallcom.2018.01.205 - DOI
-
- Chen L., Tang Y., Wang K., Liu C., Luo S. (2011). Direct electrodeposition of reduced graphene oxide on glassy carbon electrode and its electrochemical application. Electrochem. Commun. 13, 133–137. 10.1016/j.elecom.2010.11.033 - DOI
LinkOut - more resources
Full Text Sources

