Expression levels of the α7 nicotinic acetylcholine receptor in the brains of patients with Alzheimer's disease and their effect on synaptic proteins in SH-SY5Y cells
- PMID: 32582986
- PMCID: PMC7411404
- DOI: 10.3892/mmr.2020.11253
Expression levels of the α7 nicotinic acetylcholine receptor in the brains of patients with Alzheimer's disease and their effect on synaptic proteins in SH-SY5Y cells
Abstract
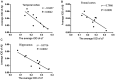
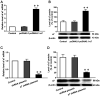
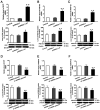
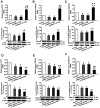
Alzheimer's disease (AD) is a chronic neurodegenerative, and abnormal aggregation of the neurotoxic β amyloid (Aβ) peptide is an early event in AD. The present study aimed to determine the correlation between the nicotinic acetylcholine receptor α7 subunit (α7 nAChR) and Aβ in the brains of patients with AD, and to investigate whether the increased expression levels of the α7 nAChR could alter the neurotoxicity of Aβ. The expression levels of α7 nAChR and Aβ in the brains of patients with AD and healthy brains were analyzed using immunofluorescence. Moreover, SH‑SY5Y cells were used to stably overexpress or silence α7 nAChR expression levels, prior to the treatment with or without 1 µmol/l Aβ1‑42 oligomer (AβO). The mRNA and protein expression levels of α7 nAChR, synaptophysin (SYP), postsynaptic density of 95 kDa (PSD‑95) and synaptosomal‑associated protein of 25 kDa (SNAP‑25) were subsequently analyzed using reverse transcription‑quantitative PCR and western blotting. In addition, the concentration of acetylcholine (ACh) and the activity of acetylcholinesterase (AChE) were analyzed using spectrophotometry, while the cell apoptotic rate was determined using flow cytometry. The expression of Aβ in the brains of patients with AD was found to be significantly increased, whereas the expression of α7 nAChR was significantly decreased compared with the healthy control group. In vitro, the expression levels of α7 nAChR were significantly increased or decreased following the overexpression or silencing of the gene, respectively. Consistent with these observations, the mRNA and protein expression levels of SYP, PSD‑95 and SNAP‑25 were also significantly increased following the overexpression of α7 nAChR and decreased following the genetic silencing of the receptor. In untransfected or negative control cells, the expression levels of these factors and the apoptotic rate were significantly reduced following the exposure to AβO, which was found to be attenuated by α7 nAChR overexpression, but potentiated by α7 nAChR RNA silencing. However, no significant differences were observed in either the ACh concentration or AChE activity following transfection. Collectively, these findings suggested that α7 nAChR may protect the brains of patients with AD against Aβ, as α7 nAChR overexpression increased the expression levels of SYP, SNAP‑25 and PSD‑95, and attenuated the inhibitory effect of Aβ on the expression of these synaptic proteins and cell apoptosis. Overall, this indicated that α7 nAChR may serve an important neuroprotective role in AD.
Keywords: SH-SY5Y cells; α7 nicotinic acetylcholine receptor; β-amyloid peptide; synaptic protein; synaptophysin; apoptotic rate; synaptosomal-associated protein of 25 kda; postsynaptic density of 95 kda; alzheimer's disease.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

