ESI-MS Study of the Interaction of Potential Oxidovanadium(IV) Drugs and Amavadin with Model Proteins
- PMID: 32585093
- PMCID: PMC8008395
- DOI: 10.1021/acs.inorgchem.0c00969
ESI-MS Study of the Interaction of Potential Oxidovanadium(IV) Drugs and Amavadin with Model Proteins
Abstract
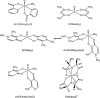
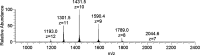
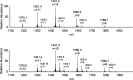
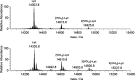
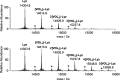
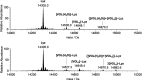
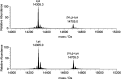
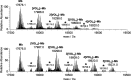
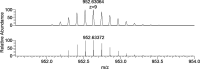
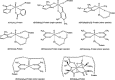
In this study, the binding to lysozyme (Lyz) of four important VIV compounds with antidiabetic and/or anticancer activity, [VIVO(pic)2(H2O)], [VIVO(ma)2], [VIVO(dhp)2], and [VIVO(acac)2], where pic-, ma-, dhp-, and acac- are picolinate, maltolate, 1,2-dimethyl-3-hydroxy-4(1H)-pyridinonate, and acetylacetonate anions, and of the vanadium-containing natural product amavadin ([VIV(hidpa)2]2-, with hidpa3- N-hydroxyimino-2,2'-diisopropionate) was investigated by ElectroSpray Ionization-Mass Spectrometry (ESI-MS). Moreover, the interaction of [VIVO(pic)2(H2O)], chosen as a representative VIVO2+ complex, was examined with two additional proteins, myoglobin (Mb) and ubiquitin (Ub), to compare the data. The examined vanadium concentration was in the range 15-150 μM, i.e., very close to that found under physiological conditions. With pic-, dhp-, and hidpa3-, the formation of adducts n[VIVOL2]-Lyz or n[VIVL2]-Lyz is favored, while with ma- and acac- the species n[VIVOL]-Lyz are detected, with n dependent on the experimental VIV/protein ratio. The behavior of the systems with [VIVO(pic)2(H2O)] and Mb or Ub is very similar to that of Lyz. The results suggested that under physiological conditions, the moiety cis-VIVOL2 (L = pic-, dhp-) is bound by only one accessible side-chain protein residue that can be Asp, Glu, or His, while VIVOL+ (L = ma-, acac-) can interact with the two equatorial and axial sites. If the VIV complex is thermodynamically stable and does not have available coordination positions, such as amavadin, the protein cannot interact with it through the formation of coordination bonds and, in such cases, noncovalent interactions are predicted. The formation of the adducts is dependent on the thermodynamic stability and geometry in aqueous solution of the VIVO2+ complex and affects the transport, uptake, and mechanism of action of potential V drugs.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




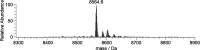

Similar articles
-
In-Depth Mass Spectrometry Study of Vanadium(IV) Complexes with Model Peptides.Inorg Chem. 2024 Sep 23;63(38):17785-17796. doi: 10.1021/acs.inorgchem.4c02683. Epub 2024 Sep 12. Inorg Chem. 2024. PMID: 39264738 Free PMC article.
-
Interaction of Vanadium(IV) Species with Ubiquitin: A Combined Instrumental and Computational Approach.Inorg Chem. 2019 Jun 17;58(12):8064-8078. doi: 10.1021/acs.inorgchem.9b00807. Epub 2019 May 29. Inorg Chem. 2019. PMID: 31140794
-
Elucidation of Binding Site and Chiral Specificity of Oxidovanadium Drugs with Lysozyme through Theoretical Calculations.Inorg Chem. 2017 Nov 6;56(21):12938-12951. doi: 10.1021/acs.inorgchem.7b01732. Epub 2017 Oct 6. Inorg Chem. 2017. PMID: 28985059
-
Interaction of antidiabetic vanadium compounds with hemoglobin and red blood cells and their distribution between plasma and erythrocytes.Inorg Chem. 2014 Feb 3;53(3):1449-64. doi: 10.1021/ic402366x. Epub 2014 Jan 17. Inorg Chem. 2014. PMID: 24437949
-
Vanadium: History, chemistry, interactions with α-amino acids and potential therapeutic applications.Coord Chem Rev. 2018 Oct 1;372:117-140. doi: 10.1016/j.ccr.2018.06.002. Epub 2018 Jun 21. Coord Chem Rev. 2018. PMID: 32226092 Free PMC article. Review.
Cited by
-
Multiple and Variable Binding of Pharmacologically Active Bis(maltolato)oxidovanadium(IV) to Lysozyme.Inorg Chem. 2022 Oct 17;61(41):16458-16467. doi: 10.1021/acs.inorgchem.2c02690. Epub 2022 Oct 7. Inorg Chem. 2022. PMID: 36205235 Free PMC article.
-
In-Depth Mass Spectrometry Study of Vanadium(IV) Complexes with Model Peptides.Inorg Chem. 2024 Sep 23;63(38):17785-17796. doi: 10.1021/acs.inorgchem.4c02683. Epub 2024 Sep 12. Inorg Chem. 2024. PMID: 39264738 Free PMC article.
-
Interaction of Vanadium Complexes with Proteins: Revisiting the Reported Structures in the Protein Data Bank (PDB) since 2015.Molecules. 2023 Sep 9;28(18):6538. doi: 10.3390/molecules28186538. Molecules. 2023. PMID: 37764313 Free PMC article. Review.
-
Speciation and structural transformation of a VV-malate complex in the absence and in the presence of a protein: from a dinuclear species to decavanadate.Inorg Chem Front. 2025 Jul 21. doi: 10.1039/d5qi01384d. Online ahead of print. Inorg Chem Front. 2025. PMID: 40757088 Free PMC article.
-
Spectroscopic/Computational Characterization and the X-ray Structure of the Adduct of the VIVO-Picolinato Complex with RNase A.Inorg Chem. 2021 Dec 20;60(24):19098-19109. doi: 10.1021/acs.inorgchem.1c02912. Epub 2021 Nov 30. Inorg Chem. 2021. PMID: 34847328 Free PMC article.
References
-
- Sakurai H.; Yoshikawa Y.; Yasui H. Current state for the development of metallopharmaceutics and anti-diabetic metal complexes. Chem. Soc. Rev. 2008, 37, 2383–2392. 10.1039/b710347f. - DOI - PubMed
- Thompson K. H.; Lichter J.; LeBel C.; Scaife M. C.; McNeill J. H.; Orvig C. Vanadium treatment of type 2 diabetes: A view to the future. J. Inorg. Biochem. 2009, 103, 554–558. 10.1016/j.jinorgbio.2008.12.003. - DOI - PubMed
- Barrio D. A.; Etcheverry S. B. Potential use of vanadium compounds in therapeutics. Curr. Med. Chem. 2010, 17, 3632–3642. 10.2174/092986710793213805. - DOI - PubMed
- Rehder D. The potentiality of vanadium in medicinal applications. Future Med. Chem. 2012, 4, 1823–1837. 10.4155/fmc.12.103. - DOI - PubMed
- Pessoa J. C.; Etcheverry S.; Gambino D. Vanadium compounds in medicine. Coord. Chem. Rev. 2015, 301–302, 24–48. 10.1016/j.ccr.2014.12.002. - DOI - PMC - PubMed
- Kioseoglou E.; Petanidis S.; Gabriel C.; Salifoglou A. The chemistry and biology of vanadium compounds in cancer therapeutics. Coord. Chem. Rev. 2015, 301-302, 87–105. 10.1016/j.ccr.2015.03.010. - DOI
- Crans D. C. Antidiabetic, Chemical, Physical Properties of Organic Vanadates as Presumed Transition-State Inhibitors for Phosphatases. J. Org. Chem. 2015, 80, 11899–11915. 10.1021/acs.joc.5b02229. - DOI - PubMed
- Rehder D. Perspectives for vanadium in health issues. Future Med. Chem. 2016, 8, 325–338. 10.4155/fmc.15.187. - DOI - PubMed
- Leon I. E.; Cadavid-Vargas J. F.; Di Virgilio A. L.; Etcheverry S. B. Vanadium, ruthenium and copper compounds: a new class of nonplatinum metallodrugs with anticancer activity. Curr. Med. Chem. 2017, 24, 112–148. 10.2174/0929867323666160824162546. - DOI - PubMed
- Crans D. C; Yang L.; Haase A.; Yang X.. Health Benefits of Vanadium and Its Potential as an Anticancer Agent. In Metal Ions in Life Sciences; Sigel A., Sigel H., Freisinger E., Sigel R. K. O., Ed.; De Gruyter GmbH: Berlin, 2018; Vol. 18, pp 251–279. - PubMed
- Crans D. C.; Henry L.; Cardiff G.; Posner B. I.. Developing Vanadium as Antidiabetic and Anticancer Drugs: A Clinical and Historical Perspective. In Essential Metals in Medicine: Therapeutic Use and Toxicity of Metal Ions in the Clinic; Carver P. L.; Ed.; De Gruyter GmbH: Berlin, 2019; Vol. 19, pp 203–230.
-
- Sanna D.; Garribba E.; Micera G. Interaction of VO2+ ion with human serum transferrin and albumin. J. Inorg. Biochem. 2009, 103, 648–655. 10.1016/j.jinorgbio.2009.01.002. - DOI - PubMed
- Sanna D.; Micera G.; Garribba E. On the Transport of Vanadium in Blood Serum. Inorg. Chem. 2009, 48, 5747–5757. 10.1021/ic802287s. - DOI - PubMed
- Sanna D.; Micera G.; Garribba E. New Developments in the Comprehension of the Biotransformation and Transport of Insulin-Enhancing Vanadium Compounds in the Blood Serum. Inorg. Chem. 2010, 49, 174–187. 10.1021/ic9017213. - DOI - PubMed
- Sanna D.; Buglyo P.; Micera G.; Garribba E. A quantitative study of the biotransformation of insulin-enhancing VO2+ compounds. JBIC, J. Biol. Inorg. Chem. 2010, 15, 825–839. 10.1007/s00775-010-0647-9. - DOI - PubMed
- Sanna D.; Micera G.; Garribba E. Interaction of VO2+ Ion and Some Insulin-Enhancing Compounds with Immunoglobulin G. Inorg. Chem. 2011, 50, 3717–3728. 10.1021/ic200087p. - DOI - PubMed
- Sanna D.; Biro L.; Buglyo P.; Micera G.; Garribba E. Biotransformation of BMOV in the presence of blood serum proteins. Metallomics 2012, 4, 33–36. 10.1039/C1MT00161B. - DOI - PubMed
- Sanna D.; Bíró L.; Buglyó P.; Micera G.; Garribba E. Transport of the anti-diabetic VO2+ complexes formed by pyrone derivatives in the blood serum. J. Inorg. Biochem. 2012, 115, 87–99. 10.1016/j.jinorgbio.2012.04.020. - DOI - PubMed
- Sanna D.; Ugone V.; Micera G.; Garribba E. Temperature and solvent structure dependence of VO2+ complexes of pyridine-N-oxide derivatives and their interaction with human serum transferrin. Dalton Trans. 2012, 41, 7304–7318. 10.1039/c2dt12503j. - DOI - PubMed
- Sanna D.; Micera G.; Garribba E. Interaction of Insulin-Enhancing Vanadium Compounds with Human Serum holo-Transferrin. Inorg. Chem. 2013, 52, 11975–11985. 10.1021/ic401716x. - DOI - PubMed
- Sanna D.; Serra M.; Micera G.; Garribba E. Interaction of Antidiabetic Vanadium Compounds with Hemoglobin and Red Blood Cells and Their Distribution between Plasma and Erythrocytes. Inorg. Chem. 2014, 53, 1449–1464. 10.1021/ic402366x. - DOI - PubMed
- Sanna D.; Serra M.; Micera G.; Garribba E. Uptake of potential anti-diabetic VIVO compounds of picolinate ligands by red blood cells. Inorg. Chim. Acta 2014, 420, 75–84. 10.1016/j.ica.2013.12.038. - DOI
- Koleša-Dobravc T.; Lodyga-Chruscinska E.; Symonowicz M.; Sanna D.; Meden A.; Perdih F.; Garribba E. Synthesis and characterization of VIVO complexes of picolinate and pyrazine derivatives. Behavior in the solid state and aqueous solution and biotransformation in the presence of blood plasma proteins. Inorg. Chem. 2014, 53, 7960–7976. 10.1021/ic500766t. - DOI - PubMed
- Sanna D.; Ugone V.; Pisano L.; Serra M.; Micera G.; Garribba E. Behavior of the potential antitumor VIVO complexes formed by flavonoid ligands. 2. Characterization of sulfonate derivatives of quercetin and morin, interaction with the bioligands of the plasma and preliminary biotransformation studies. J. Inorg. Biochem. 2015, 153, 167–177. 10.1016/j.jinorgbio.2015.07.018. - DOI - PubMed
- Sanna D.; Ugone V.; Micera G.; Pivetta T.; Valletta E.; Garribba E. Speciation of the Potential Antitumor Agent Vanadocene Dichloride in the Blood Plasma and Model Systems. Inorg. Chem. 2015, 54, 8237–8250. 10.1021/acs.inorgchem.5b01277. - DOI - PubMed
- Sanna D.; Ugone V.; Sciortino G.; Buglyo P.; Bihari Z.; Parajdi-Losonczi P. L.; Garribba E. VIVO complexes with antibacterial quinolone ligands and their interaction with serum proteins. Dalton Trans. 2018, 47, 2164–2182. 10.1039/C7DT04216G. - DOI - PubMed
- Sciortino G.; Sanna D.; Ugone V.; Maréchal J.-D.; Alemany-Chavarria M.; Garribba E. Effect of secondary interactions, steric hindrance and electric charge on the interaction of VIVO species with proteins. New J. Chem. 2019, 43, 17647–17660. 10.1039/C9NJ01956A. - DOI
-
- Willsky G. R.; Goldfine A. B.; Kostyniak P. J.; McNeill J. H.; Yang L. Q.; Khan H. R.; Crans D. C. Effect of vanadium(IV) compounds in the treatment of diabetes: in vivo and in vitro studies with vanadyl sulfate and bis(maltolato)oxovandium(IV). J. Inorg. Biochem. 2001, 85, 33–42. 10.1016/S0162-0134(00)00226-9. - DOI - PubMed
- Liboiron B. D.; Thompson K. H.; Hanson G. R.; Lam E.; Aebischer N.; Orvig C. New Insights into the Interactions of Serum Proteins with Bis(maltolato)oxovanadium(IV): Transport and Biotransformation of Insulin-Enhancing Vanadium Pharmaceuticals. J. Am. Chem. Soc. 2005, 127, 5104–5115. 10.1021/ja043944n. - DOI - PubMed
- Jakusch T.; Hollender D.; Enyedy E. A.; Gonzalez C. S.; Montes-Bayon M.; Sanz-Medel A.; Pessoa J. C.; Tomaz I.; Kiss T. Biospeciation of various antidiabetic VIVO compounds in serum. Dalton Trans. 2009, 2428–2437. 10.1039/b817748a. - DOI - PubMed
- Correia I.; Jakusch T.; Cobbinna E.; Mehtab S.; Tomaz I.; Nagy N. V.; Rockenbauer A.; Pessoa J. C.; Kiss T. Evaluation of the binding of oxovanadium(IV) to human serum albumin. Dalton Trans. 2012, 41, 6477–6487. 10.1039/c2dt12193j. - DOI - PubMed
- Mehtab S.; Goncalves G.; Roy S.; Tomaz A. I.; Santos-Silva T.; Santos M. F. A.; Romao M. J.; Jakusch T.; Kiss T.; Pessoa J. C. Interaction of vanadium(IV) with human serum apo-transferrin. J. Inorg. Biochem. 2013, 121, 187–195. 10.1016/j.jinorgbio.2012.12.020. - DOI - PubMed
- Goncalves G.; Tomaz I.; Correia I.; Veiros L. F.; Castro M. M. C. A.; Avecilla F.; Palacio L.; Maestro M.; Kiss T.; Jakusch T.; Garcia M. H. V.; Pessoa J. C. A novel VIVO-pyrimidinone complex: synthesis, solution speciation and human serum protein binding. Dalton Trans. 2013, 42, 11841–11861. 10.1039/c3dt50553g. - DOI - PubMed
- Justino G. C.; Garribba E.; Pessoa J. C. Binding of VIVO2+ to the Fe binding sites of human serum transferrin. A theoretical study. JBIC, J. Biol. Inorg. Chem. 2013, 18, 803–813. 10.1007/s00775-013-1029-x. - DOI - PubMed
- Santos M. F. A.; Correia I.; Oliveira A. R.; Garribba E.; Pessoa J. C.; Santos-Silva T. Vanadium complexes as prospective therapeutics: Structural characterization of a VIV lysozyme adduct. Eur. J. Inorg. Chem. 2014, 2014, 3293–3297. 10.1002/ejic.201402408. - DOI
- Pessoa J. C.; Gonçalves G.; Roy S.; Correia I.; Mehtab S.; Santos M. F. A.; Santos-Silva T. New insights on vanadium binding to human serum transferrin. Inorg. Chim. Acta 2014, 420, 60–68. 10.1016/j.ica.2013.11.025. - DOI
- Pessoa J. C. Thirty years through vanadium chemistry. J. Inorg. Biochem. 2015, 147, 4–24. 10.1016/j.jinorgbio.2015.03.004. - DOI - PubMed
- Pessoa J. C.; Garribba E.; Santos M. F. A.; Santos-Silva T. Vanadium and proteins: uptake, transport, structure, activity and function. Coord. Chem. Rev. 2015, 301–302, 49–86. 10.1016/j.ccr.2015.03.016. - DOI
- Correia I.; Chorna I.; Cavaco I.; Roy S.; Kuznetsov M. L.; Ribeiro N.; Justino G.; Marques F.; Santos-Silva T.; Santos M. F. A.; Santos H. M.; Capelo J. L.; Doutch J.; Pessoa J. C. Interaction of [VIVO(acac)2] with human serum transferrin and albumin. Chem. - Asian J. 2017, 12, 2062–2084. 10.1002/asia.201700469. - DOI - PubMed
- Jakusch T.; Kiss T. In vitro study of the antidiabetic behavior of vanadium compounds. Coord. Chem. Rev. 2017, 351, 118–126. 10.1016/j.ccr.2017.04.007. - DOI
- Levina A.; Crans D. C.; Lay P. A. Speciation of metal drugs, supplements and toxins in media and bodily fluids controls in vitro activities. Coord. Chem. Rev. 2017, 352, 473–498. 10.1016/j.ccr.2017.01.002. - DOI
- Azevedo C. G.; Correia I.; dos Santos M. M. C.; Santos M. F. A.; Santos-Silva T.; Doutch J.; Fernandes L.; Santos H. M.; Capelo J. L.; Pessoa J. C. Binding of vanadium to human serum transferrin - voltammetric and spectrometric studies. J. Inorg. Biochem. 2018, 180, 211–221. 10.1016/j.jinorgbio.2017.12.012. - DOI - PubMed
-
- Rehder D.Bioinorganic Vanadium Chemistry; John Wiley & Sons Ltd.: Chichester, 2008.
-
- Boukhobza I.; Crans D. C. Application of HPLC to measure vanadium in environmental, biological and clinical matrices. Arabian J. Chem. 2020, 13, 1198–1228. 10.1016/j.arabjc.2017.10.003. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

