Persistence with Anticoagulation for Atrial Fibrillation: Report from the GLORIA-AF Phase III 1-Year Follow-up
- PMID: 32586056
- PMCID: PMC7356563
- DOI: 10.3390/jcm9061969
Persistence with Anticoagulation for Atrial Fibrillation: Report from the GLORIA-AF Phase III 1-Year Follow-up
Abstract
Background: We aimed to assess the extent to which drug persistence is better with non-vitamin K antagonist oral anticoagulants (NOACs) than vitamin K antagonists (VKAs) in atrial fibrillation (AF) patients and to estimate the difference in therapy persistence depending on NOAC dosing regimen (once daily (QD) vs. twice daily (BID)).
Methods: Consecutive patients were followed for 1 year in phase III of the GLORIA-AF registry. Drug persistence was defined as the use of OAC without any discontinuation in >30 days or switching to alternative therapy.
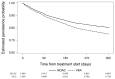
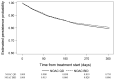
Results: Among 21,109 eligible patients in phase III, 17,266 patients who were prescribed OAC at baseline and those who took ≥1 OAC dose were included. The 1-year proportion of patients receiving NOAC and VKA who persisted on treatment was 80% and 75%, respectively. The 1-year persistence with NOACs BID and NOACs QD was 81% and 80%, respectively. Female gender, hypertension, older age, alcohol use, permanent, asymptomatic, and minimally symptomatic AF were associated with better OAC persistence. Region, medication usage predisposing to bleeding, being a current smoker, treatment reimbursement, and proton pump inhibitors were associated with lower OAC persistence.
Conclusions: Drug persistence was higher with NOACs (1-year persistence was 80%) than with VKAs (75%). There was little difference in 1-year persistence between NOAC dosing regimens.
Keywords: GLORIA-AF; atrial fibrillation; dosing frequency; non-vitamin K antagonist oral anticoagulants; oral anticoagulants; vitamin K antagonists.
Conflict of interest statement
Kozieł, Mazurek, and Rothman declare no conflicts of interest. Teutsch, Paquette, Zint, and Lu are employees of Boehringer Ingelheim. Bartels and Riou Franca were employees of Boehringer Ingelheim at the time of manuscript writing. Diener received honoraria for participation in clinical trials, contribution to advisory boards, or oral presentations from Abbott, Bayer Vital, BMS, Boehringer Ingelheim, Daiichi-Sankyo, Medtronic, Pfizer, Portola, Sanofi-Aventis, and WebMD Global. Financial support for research projects was provided by Boehringer Ingelheim. HCD chairs the Treatment Guidelines Committee of the German Society of Neurology and contributed to the EHRA and ESC guidelines for the treatment of AF. Dubner has received consultancy fees for serving as a steering committee member for Boehringer Ingelheim. He also holds research grants from Abbott. Halperin has engaged in consulting activities with Boehringer Ingelheim for advisory activities involving anticoagulants, and he is a member of the Executive Steering Committee of the GLORIA-AF Registry. Ma has received honoraria for lectures from AstraZeneca, Bayer HealthCare, Boehringer Ingelheim, Bristol-Myers Squibb, Johnson & Johnson, and Pfizer. Brandes has received lecture fees from Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, and MSD and research grants from Gilead and the Region of Southern Denmark and Zealand. Huisman reports grants from ZonMW Dutch Healthcare Fund, grants and personal fees from Boehringer Ingelheim, Pfizer-BMS, Bayer HealthCare, Aspen, and Daiichi-Sankyo, outside the submitted work. Lip has been a consultant for Bayer/Janssen, BMS/Pfizer, Medtronic, Boehringer Ingelheim, Novartis, Verseon, and Daiichi-Sankyo. He has been a speaker for Bayer, BMS/Pfizer, Medtronic, Boehringer Ingelheim, and Daiichi-Sankyo. He did not directly receive any fees.
Figures
Similar articles
-
Antithrombotic therapy use in patients with atrial fibrillation before the era of non-vitamin K antagonist oral anticoagulants: the Global Registry on Long-Term Oral Antithrombotic Treatment in Patients with Atrial Fibrillation (GLORIA-AF) Phase I cohort.Europace. 2016 Sep;18(9):1308-18. doi: 10.1093/europace/euw073. Epub 2016 Jun 21. Europace. 2016. PMID: 27335063 Free PMC article.
-
Evolution of antithrombotic therapy for patients with atrial fibrillation: The prospective global GLORIA-AF registry program.PLoS One. 2022 Oct 6;17(10):e0274237. doi: 10.1371/journal.pone.0274237. eCollection 2022. PLoS One. 2022. PMID: 36201473 Free PMC article.
-
Adherence and persistence to oral anticoagulants in patients with atrial fibrillation: A Belgian nationwide cohort study.Front Cardiovasc Med. 2022 Sep 29;9:994085. doi: 10.3389/fcvm.2022.994085. eCollection 2022. Front Cardiovasc Med. 2022. PMID: 36247477 Free PMC article.
-
The 2018 European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation: executive summary.Europace. 2018 Aug 1;20(8):1231-1242. doi: 10.1093/europace/euy054. Europace. 2018. PMID: 29562331 Review.
-
Appropriate doses of non-vitamin K antagonist oral anticoagulants in high-risk subgroups with atrial fibrillation: Systematic review and meta-analysis.J Cardiol. 2018 Oct;72(4):284-291. doi: 10.1016/j.jjcc.2018.03.009. Epub 2018 Apr 26. J Cardiol. 2018. PMID: 29706404
Cited by
-
Non‑persistence to Oral Anticoagulation Therapy in Elderly Patients with Non‑valvular Atrial Fibrillation.Patient Prefer Adherence. 2023 Dec 6;17:3185-3194. doi: 10.2147/PPA.S435592. eCollection 2023. Patient Prefer Adherence. 2023. PMID: 38084322 Free PMC article.
-
Outcomes in elderly Chinese patients with atrial fibrillation and coronary artery disease. A report from the Optimal Thromboprophylaxis in Elderly Chinese Patients with Atrial Fibrillation (ChiOTEAF) registry.J Arrhythm. 2022 May 31;38(4):580-588. doi: 10.1002/joa3.12744. eCollection 2022 Aug. J Arrhythm. 2022. PMID: 35936042 Free PMC article.
-
The temporal context of oral anticoagulation outcome in atrial fibrillation.Int J Cardiol Heart Vasc. 2022 May 13;40:101051. doi: 10.1016/j.ijcha.2022.101051. eCollection 2022 Jun. Int J Cardiol Heart Vasc. 2022. PMID: 35663453 Free PMC article. No abstract available.
-
Poor adherence to guideline-directed anticoagulation in elderly Chinese patients with atrial fibrillation: a report from the Optimal Thromboprophylaxis in Elderly Chinese Patients with Atrial Fibrillation (ChiOTEAF) registry.Eur Heart J Qual Care Clin Outcomes. 2023 Feb 28;9(2):169-176. doi: 10.1093/ehjqcco/qcab054. Eur Heart J Qual Care Clin Outcomes. 2023. PMID: 34370024 Free PMC article.
-
Optimizing adherence and persistence to non-vitamin K antagonist oral anticoagulant therapy in atrial fibrillation.Eur Heart J Suppl. 2022 Feb 14;24(Suppl A):A42-A55. doi: 10.1093/eurheartj/suab152. eCollection 2022 Feb. Eur Heart J Suppl. 2022. PMID: 35185408 Free PMC article.
References
-
- Lip G., Freedman B., De Caterina R., Potpara T.S. Stroke prevention in atrial fibrillationast, present and future. Comparing the guidelines and practical decision-making. Thromb. Haemost. 2017;117:1230–1239. - PubMed
-
- Paquette M., França L.R., Teutsch C., Diener H.-C., Lu S., Dubner S.J., Rothman J.K., Zint K., Halperin J.L., Huisman M.V., et al. Persistence With Dabigatran Therapy at 2 Years in Patients with Atrial Fibrillation. J. Am. Coll. Cardiol. 2017;70:1573–1583. doi: 10.1016/j.jacc.2017.07.793. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

