Tumor Mutational Burden, Toxicity, and Response of Immune Checkpoint Inhibitors Targeting PD(L)1, CTLA-4, and Combination: A Meta-regression Analysis
- PMID: 32586938
- PMCID: PMC7501151
- DOI: 10.1158/1078-0432.CCR-20-0458
Tumor Mutational Burden, Toxicity, and Response of Immune Checkpoint Inhibitors Targeting PD(L)1, CTLA-4, and Combination: A Meta-regression Analysis
Abstract
Purpose: Tumor mutational burden (TMB) has emerged as a potential predictive biomarker for clinical response to ICI therapy, but whether TMB also predicts toxicity remains unknown. We investigated the relationship between TMB, objective response rate (ORR), overall survival (OS), and toxicity for ICI therapy across multiple cancer types.
Experimental design: We searched MEDLINE, PubMed, and ASCO/ESMO/AACR meetings for clinical trials of anti-PD(L)1, CTLA-4, or combination in 29 cancer types. We assessed ICI administered, responses (complete or partial response), median OS, OS HR, and grade 3/4 toxicity. We conducted a systematic review, meta-analysis and meta-regression using tumor level TMB data from Foundation Medicine.
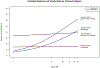
Results: One hundred seventeen clinical trials, which included 12,450 patients treated with ICI therapy were analyzed. Meta-regression analysis revealed that TMB was significantly associated with ORR for anti-PD(L)1, anti-CTLA-4, and combination (P < 0.0001 for all), but not associated with toxicity in all treatment groups. OS data were unavailable for most studies included in our meta-analysis, and the relationship between TMB and OS in this subset was not significant (P = 0.26). In high TMB tumor types (≥10 mut/megabase) the improvement of ORR and increase in grade 3/4 toxicity with combination ICI therapy as compared with PD(L)1 monotherapy were 21.13% and 25.41%, respectively, as compared with 3.73% and 18.78% in low TMB tumor types (<10 mut/megabase).
Conclusions: There is a positive association between TMB and clinical response with anti-PD(L)1, anti-CTLA-4, and combination ICIs, but no association between TMB and toxicity. These results imply a favorable risk/benefit ratio for ICIs in tumors with a higher TMB.
©2020 American Association for Cancer Research.
Conflict of interest statement
Figures




References
-
- Paz-Ares L, Luft A, Vicente D, et al. Pembrolizumab plus Chemotherapy for Squamous Non–Small-Cell Lung Cancer. N Engl J Med 2018; 379: 2040–51. - PubMed
-
- Osipov A, Murphy A, Zheng L. From immune checkpoints to vaccines: The past, present and future of cancer immunotherapy In: Advances in Cancer Research. Academic Press, 2019: 63–144. - PubMed
-
- Spencer KR, Wang J, Silk AW, Ganesan S, Kaufman HL, Mehnert JM. Biomarkers for Immunotherapy: Current Developments and Challenges. Am Soc Clin Oncol Educ B 2016; 36: e493–503. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

