Boosting of the enhanced permeability and retention effect with nanocapsules improves the therapeutic effects of cetuximab
- PMID: 32587779
- PMCID: PMC7309461
- DOI: 10.20892/j.issn.2095-3941.2019.0292
Boosting of the enhanced permeability and retention effect with nanocapsules improves the therapeutic effects of cetuximab
Abstract
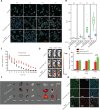
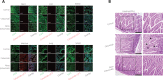
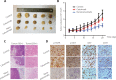
Objective: The introduction of therapeutic antibodies (tAbs) into clinical practice has revolutionized tumor treatment strategies, but their tumor therapy efficiency is still far below expectations because of the rapid degradation and limited tumor accumulation of tAbs. Methods: We developed a nanocapsule-based delivery system to induce the self-augmentation of the enhanced permeability and retention (EPR) effect. This system constantly penetrated across the blood-tumor barrier into the tumor while avoiding the attack of tAbs by the immune system. The biodistribution and therapeutic effect were tested with single dose administration of nanocapsule-tAbs in vivo. Results: The accumulation of Nano(cetuximab) within subcutaneous PC9 tumors was gradually enhanced over 6 days after single dose administration, which was contrary to the biodistribution of native cetuximab. Nano(cetuximab) accumulated in tumor tissues via the EPR effect and released cetuximab. The released cetuximab acted on vascular endothelial cells to destroy the blood-tumor barrier and induce self-augmentation of the EPR effect, which in turn contributed to further tumor accumulation of long-circulating Nano(cetuximab). Compared with single dose administration of native cetuximab, Nano(cetuximab) showed an effective tumor suppressive effect for 3 weeks. Conclusions: The nanocapsule-based delivery system efficiently delivered tAbs to tumor tissues and released them to boost the EPR effect, which facilitated further tumor accumulation of the tAbs. This novel self-augmentation of the EPR effect facilitated by the biological characteristics of tAbs and nanotechnology contributed to the improvement of the therapeutic effect of tAbs, and stimulated new ideas for antibody-based tumor therapy.
Keywords: EPR effect; Endothelial cells; nanocapsule; single dose administration; therapeutic antibody.
Copyright: © 2020, Cancer Biology & Medicine.
Conflict of interest statement
*These authors contributed equally to this work.
Figures


References
-
- Meulendijks D, Jacob W, Voest EE, Mau-Sorensen M, Martinez-Garcia M, Taus A, et al. Phase Ib study of lumretuzumab plus cetuximab or erlotinib in solid tumor patients and evaluation of HER3 and heregulin as potential biomarkers of clinical activity. Clin Cancer Res. 2017;23:5406–15. - PubMed
-
- Hewish M, Cunningham D. First-line treatment of advanced colorectal cancer. Lancet. 2011;377:2060–2. - PubMed
-
- Ciardiello F, Tortora G. EGFR antagonists in cancer treatment. N Engl J Med. 2008;358:1160–74. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
