Recognition of Semaphorin Proteins by P. sordellii Lethal Toxin Reveals Principles of Receptor Specificity in Clostridial Toxins
- PMID: 32589945
- PMCID: PMC7316060
- DOI: 10.1016/j.cell.2020.06.005
Recognition of Semaphorin Proteins by P. sordellii Lethal Toxin Reveals Principles of Receptor Specificity in Clostridial Toxins
Abstract
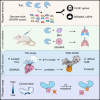
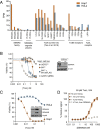
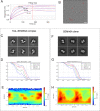
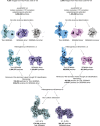
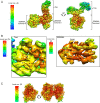
Pathogenic clostridial species secrete potent toxins that induce severe host tissue damage. Paeniclostridium sordellii lethal toxin (TcsL) causes an almost invariably lethal toxic shock syndrome associated with gynecological infections. TcsL is 87% similar to C. difficile TcdB, which enters host cells via Frizzled receptors in colon epithelium. However, P. sordellii infections target vascular endothelium, suggesting that TcsL exploits another receptor. Here, using CRISPR/Cas9 screening, we establish semaphorins SEMA6A and SEMA6B as TcsL receptors. We demonstrate that recombinant SEMA6A can protect mice from TcsL-induced edema. A 3.3 Å cryo-EM structure shows that TcsL binds SEMA6A with the same region that in TcdB binds structurally unrelated Frizzled. Remarkably, 15 mutations in this evolutionarily divergent surface are sufficient to switch binding specificity of TcsL to that of TcdB. Our findings establish semaphorins as physiologically relevant receptors for TcsL and reveal the molecular basis for the difference in tissue targeting and disease pathogenesis between highly related toxins.
Keywords: CRISPR/Cas9 screening; Clostridial toxins; P. sordellii; SEMA6A; SEMA6B; TcsL; bacterial exotoxins; cryo-EM; semaphorin.
Copyright © 2020 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Interests The authors declare no competing interests.
Figures

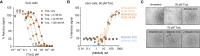



Comment in
-
Semaphorins or Frizzled -it is the receptor that direct the action of clostridial glucosylating toxins.Signal Transduct Target Ther. 2020 Sep 19;5(1):206. doi: 10.1038/s41392-020-00307-3. Signal Transduct Target Ther. 2020. PMID: 32951001 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

