Non-Myeloablative Allogeneic Transplantation with Post-Transplant Cyclophosphamide after Immune Checkpoint Inhibition for Classic Hodgkin Lymphoma: A Retrospective Cohort Study
- PMID: 32592857
- PMCID: PMC7486273
- DOI: 10.1016/j.bbmt.2020.06.012
Non-Myeloablative Allogeneic Transplantation with Post-Transplant Cyclophosphamide after Immune Checkpoint Inhibition for Classic Hodgkin Lymphoma: A Retrospective Cohort Study
Abstract
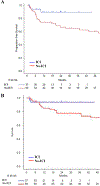
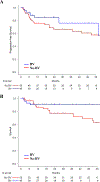
: Immune checkpoint inhibitors (ICIs) are approved in relapsed classic Hodgkin lymphoma (cHL). The safety and effectiveness of allogeneic blood or marrow transplantation (alloBMT) in ICI-pretreated patients with cHL remain unclear. The aim of this study is to assess outcomes of patients with cHL receiving ICIs before alloBMT using post-transplantation cyclophosphamide (PTCy) graft-versus-host-disease (GVHD) prophylaxis. : We performed a retrospective study of relapsed/refractory patients with cHL undergoing alloBMT with PTCy at Johns Hopkins between November 2004 and September 2019. Engraftment, GVHD incidence, nonrelapse mortality, progression-free survival (PFS), and overall survival (OS) were compared between patients receiving pre-alloBMT ICI or standard salvage chemotherapy. : We identified 105 consecutive relapsed/refractory patients with cHL, of whom 37 (35.2%) received ICIs and 68 (64.7%) received chemotherapy without ICIs (no-ICI) before alloBMT. ICI and no-ICI patients experienced a 3-year estimated OS of 94% versus 78% (hazard ratio [HR], 0.35; 95% confidence interval [CI], 0.08 to 1.56; P = .17) and a 3-year estimated PFS of 90% and 65% (HR, 0.3; 95% CI, 0.09 to 1; P = .05), respectively. We observed no statically significant difference in the 12-month cumulative incidence of acute grade II to IV GVHD or in the 24-month incidence of chronic GVHD. : ICIs do not increase acute or chronic GVHD incidence compared with salvage chemotherapy. Patients with cHL receiving ICIs prior to alloBMT experienced outstanding PFS and OS. Thus, ICI therapy is safe in patients with cHL when undergoing alloBMT with PTCy and may improve post-alloBMT disease progression and survival.
Keywords: Allogeneic blood or marrow transplantation (alloBMT); Classic Hodgkin lymphoma (cHL); Engraftment; Graft-versus-host-disease (GVHD); Immune checkpoint Inhibitors (ICIs); Post-transplantation cyclophosphamide (PTCy).
Copyright © 2020 American Society for Transplantation and Cellular Therapy. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest statements
N.W.J participates in advisory boards of Regeneron, ADC Therapeutics, CALIBR, Versatem, Bayer, Gilead. J.B.M has received honoraria from Incyte Corporation as a member of their Data Monitoring Committee.
Ethics committee approval
Study was approved by Johns Hopkins Medicine Institutional Review Board.
Figures



References
-
- Castagna L, Bramanti S, Devillier R, et al. Haploidentical transplantation with post-infusion cyclophosphamide in advanced Hodgkin lymphoma. Bone marrow transplantation 2017; 52(5): 683–8. - PubMed
-
- Mariotti J, Devillier R, Bramanti S, et al. T Cell-Replete Haploidentical Transplantation with Post-Transplantation Cyclophosphamide for Hodgkin Lymphoma Relapsed after Autologous Transplantation: Reduced Incidence of Relapse and of Chronic Graft-versus-Host Disease Compared with HLA-Identical Related Donors. Biology of blood and marrow transplantation : journal of the American Society for Blood and Marrow Transplantation 2018; 24(3): 627–32. - PubMed
-
- Martinez C, Gayoso J, Canals C, et al. Post-Transplantation Cyclophosphamide-Based Haploidentical Transplantation as Alternative to Matched Sibling or Unrelated Donor Transplantation for Hodgkin Lymphoma: A Registry Study of the Lymphoma Working Party of the European Society for Blood and Marrow Transplantation. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2017; 35(30): 3425–32. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

