Photoprotective Effects of Selected Amino Acids on Naproxen Photodegradation in Aqueous Media
- PMID: 32604908
- PMCID: PMC7345999
- DOI: 10.3390/ph13060135
Photoprotective Effects of Selected Amino Acids on Naproxen Photodegradation in Aqueous Media
Abstract
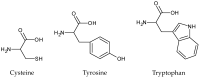
It is important to develop a photostabilization strategy to ensure the quality of photosensitive compounds, including pharmaceuticals. This study focused on the protective effects of 20 amino acids on the photodegradation of naproxen (NX), a photosensitive pharmaceutical, to clarify the important nature of a good photostabilizer. Our previous report indicated the photodegradability of NX and the protective effects of some antioxidants on its photodegradation, therefore, this compound was used as a model compound. The degradation of NX in aqueous media during ultraviolet light (UV) irradiation and the protective effects of selected amino acids were monitored through high-performance liquid chromatography (HPLC), equipped with a reverse-phase column. Addition of cysteine, tryptophan, and tyrosine induced the significant suppression of NX photodegradation after UV irradiation for 3 h (residual amount of NX; 15.35%, 6.82%, and 15.64%, respectively). Evaluation of the antioxidative activity and UV absorption spectrum showed that cysteine suppressed NX degradation through its antioxidative ability, while tryptophan and tyrosine suppressed it through their UV filtering ability. Furthermore, three amino acids at higher concentrations (more than 100 µmol/L) showed more protective effects on NX photodegradation. For 10 mmol/L, residual amounts of NX with cysteine, tryptophan, and tyrosine were 58.51%, 69.34%, and 82.40%, respectively. These results showed the importance of both photoprotective potencies (antioxidative potency and UV filtering potency) and stability to UV irradiation for a good photostabilizer of photosensitive pharmaceuticals.
Keywords: HPLC; UV/Vis spectroscopy; analytical chemistry; antioxidative activity; chromatography; photochemistry; photodegradation; photostability; photostabilization.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Viola G., Grobelny P., Linardi M.A., Salvador A., Basso G., Mielcarek J., Dall’Acqua S., Vedaldi D., Dall’Acqua F. The phototoxicity of fluvastatin, an HMG-CoA reductase inhibitor, is mediated by the formation of a benzocarbazole-like photoproduct. Toxicol. Sci. 2010;118:236–250. doi: 10.1093/toxsci/kfq228. - DOI - PubMed
-
- Kawabata K., Mizuta Y., Ishihara K., Takato A., Oshima S., Akimoto S., Inagaki M., Nishi H. Structure determination of naproxen photoproducts in the tablet generated by the UV irradiation. Chromatography. 2019;40:157–162. doi: 10.15583/jpchrom.2019.020. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

