Viral Membrane Fusion and the Transmembrane Domain
- PMID: 32604992
- PMCID: PMC7412173
- DOI: 10.3390/v12070693
Viral Membrane Fusion and the Transmembrane Domain
Abstract
Initiation of host cell infection by an enveloped virus requires a viral-to-host cell membrane fusion event. This event is mediated by at least one viral transmembrane glycoprotein, termed the fusion protein, which is a key therapeutic target. Viral fusion proteins have been studied for decades, and numerous critical insights into their function have been elucidated. However, the transmembrane region remains one of the most poorly understood facets of these proteins. In the past ten years, the field has made significant advances in understanding the role of the membrane-spanning region of viral fusion proteins. We summarize developments made in the past decade that have contributed to the understanding of the transmembrane region of viral fusion proteins, highlighting not only their critical role in the membrane fusion process, but further demonstrating their involvement in several aspects of the viral lifecycle.
Keywords: transmembrane domain; viral fusion protein.
Conflict of interest statement
The authors report no conflict of interest.
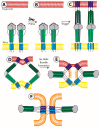
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

