Obesity/Type 2 Diabetes-Associated Liver Tumors Are Sensitive to Cyclin D1 Deficiency
- PMID: 32606000
- PMCID: PMC7442681
- DOI: 10.1158/0008-5472.CAN-20-0106
Obesity/Type 2 Diabetes-Associated Liver Tumors Are Sensitive to Cyclin D1 Deficiency
Abstract
Type 2 diabetes, which is mainly linked to obesity, is associated with increased incidence of liver cancer. We have previously found that in various models of obesity/diabetes, hyperinsulinemia maintains heightened hepatic expression of cyclin D1, suggesting a plausible mechanism linking diabetes and liver cancer progression. Here we show that cyclin D1 is greatly elevated in human livers with diabetes and is among the most significantly upregulated genes in obese/diabetic liver tumors. Liver-specific cyclin D1 deficiency protected obese/diabetic mice against hepatic tumorigenesis, whereas lean/nondiabetic mice developed tumors irrespective of cyclin D1 status. Cyclin D1 dependency positively correlated with liver cancer sensitivity to palbociclib, an FDA-approved CDK4 inhibitor, which was effective in treating orthotopic liver tumors under obese/diabetic conditions. The antidiabetic drug metformin suppressed insulin-induced hepatic cyclin D1 expression and protected against obese/diabetic hepatocarcinogenesis. These results indicate that the cyclin D1-CDK4 complex represents a potential selective therapeutic vulnerability for liver tumors in obese/diabetic patients. SIGNIFICANCE: Obesity/diabetes-associated liver tumors are specifically vulnerable to cyclin D1 deficiency and CDK4 inhibition, suggesting that the obese/diabetic environment confers cancer-selective dependencies that can be therapeutically exploited.
©2020 American Association for Cancer Research.
Conflict of interest statement
Declaration of Interests
The authors declare no competing interests.
Figures

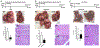


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

