Effect of Intravenous Lidocaine on Postoperative Pain in Patients Undergoing Intraspinal Tumor Resection: Study Protocol for a Prospective Randomized Controlled Trial
- PMID: 32606906
- PMCID: PMC7297458
- DOI: 10.2147/JPR.S249359
Effect of Intravenous Lidocaine on Postoperative Pain in Patients Undergoing Intraspinal Tumor Resection: Study Protocol for a Prospective Randomized Controlled Trial
Abstract
Purpose: Patients undergoing intraspinal tumor resection usually experience severe acute pain, delaying postoperative rehabilitation, and increasing incidence of chronic pain. Recently, an increasing number of studies have found that low-dose intravenous lidocaine infusion during and/or after surgery can reduce opioid usage and the incidence of related side effects, inhibit hyperalgesia and promote recovery. Thus far, no studies have evaluated the analgesic effect and safety of perioperative intravenous lidocaine infusion for intraspinal tumor resection, especially the long-term analgesic effects of patient-controlled analgesia (PCA) with lidocaine during the first postoperative 48 hours. This study tests the hypothesis that intra- and postoperative systemic lidocaine infusion for patients undergoing intraspinal tumor resection can relieve postoperative acute or chronic pain and reduce the opioid dosage and incidence of related side effects without other problems.
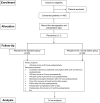
Study design and methods: This is a prospective, randomized, placebo-controlled, and double-blinded study. In total, 180 participants scheduled for intraspinal tumor resection will be randomly divided into lidocaine and placebo groups. The lidocaine group will be administered lidocaine intravenously during anesthesia and postoperative pain management during the first 48 postoperative hours; the placebo group will be administered normal saline at the same volume, infusion rate, and timing. The primary outcome will be the postoperative visual analog scale (VAS) score. Secondary outcomes will be postoperative cumulative sufentanil consumption, indicators of postoperative recovery, and the incidence of perioperative adverse events.
Discussion: This study investigates the effect of continuous intravenous lidocaine infusion on postoperative sufentanil consumption and VAS scores. The findings will provide a new strategy of anesthesia and analgesia management for intraspinal tumor resection.
Keywords: intraspinal tumor resection; lidocaine; opiates; postoperative pain.
© 2020 Yue et al.
Conflict of interest statement
The authors report no conflicts of interest related to this work.
Figures
Similar articles
-
Effects of Lidocaine on Motor-Evoked Potentials and Somatosensory-Evoked Potentials in Patients Undergoing Intraspinal Tumour Resection: Study Protocol for a Prospective Randomized Controlled Trial.J Pain Res. 2022 Feb 2;15:287-297. doi: 10.2147/JPR.S345091. eCollection 2022. J Pain Res. 2022. PMID: 35140514 Free PMC article.
-
Effect of Sufentanil Combined with Gabapentin on Acute Postoperative Pain in Patients Undergoing Intraspinal Tumor Resection: Study Protocol for a Randomized Controlled Trial.J Pain Res. 2022 Sep 1;15:2619-2628. doi: 10.2147/JPR.S374898. eCollection 2022. J Pain Res. 2022. PMID: 36072908 Free PMC article.
-
Efficacy of Perioperative Continuous Intravenous Lidocaine Infusion for 72 Hours on Postoperative Pain and Recovery in Patients Undergoing Hepatectomy: Study Protocol for a Prospective Randomized Controlled Trial.J Pain Res. 2021 Dec 1;14:3665-3674. doi: 10.2147/JPR.S341550. eCollection 2021. J Pain Res. 2021. PMID: 34880671 Free PMC article.
-
Perioperative Intravenous Lidocaine Infusion for Postoperative Analgesia in Patients Undergoing Surgery of the Spine: Systematic Review and Meta-Analysis.Pain Med. 2022 Jan 3;23(1):45-56. doi: 10.1093/pm/pnab210. Pain Med. 2022. PMID: 34196720
-
Perioperative intravenous lidocaine for postoperative pain in patients undergoing breast surgery: a meta-analysis with trial sequential analysis of randomized controlled trials.Front Oncol. 2023 Jun 23;13:1101582. doi: 10.3389/fonc.2023.1101582. eCollection 2023. Front Oncol. 2023. PMID: 37427130 Free PMC article.
Cited by
-
Effects of Lidocaine on Motor-Evoked Potentials and Somatosensory-Evoked Potentials in Patients Undergoing Intraspinal Tumour Resection: Study Protocol for a Prospective Randomized Controlled Trial.J Pain Res. 2022 Feb 2;15:287-297. doi: 10.2147/JPR.S345091. eCollection 2022. J Pain Res. 2022. PMID: 35140514 Free PMC article.
References
LinkOut - more resources
Full Text Sources