Synergistic Anticancer Effects of Gemcitabine with Pitavastatin on Pancreatic Cancer Cell Line MIA PaCa-2 in vitro and in vivo
- PMID: 32606957
- PMCID: PMC7306478
- DOI: 10.2147/CMAR.S247876
Synergistic Anticancer Effects of Gemcitabine with Pitavastatin on Pancreatic Cancer Cell Line MIA PaCa-2 in vitro and in vivo
Abstract
Background: Pancreatic ductal adenocarcinoma (PDAC) is a highly aggressive malignancy with an overall 5-year survival rate of 9.3%, and this malignancy is expected to become the second leading cause of cancer-related death by 2030. Gemcitabine resistance develops within weeks of PDAC patient's chemotherapeutic initiation. Statins, including pitavastatin, have been indicated to have anticancer effects in numerous human cancer cell lines. Thus, in this study, we hypothesized that a combination of gemcitabine and pitavastatin may have a greater anticancer effect than gemcitabine alone on the human pancreatic carcinoma cell line MIA PaCa-2.
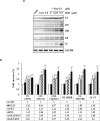
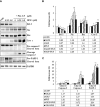
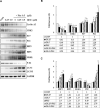
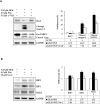
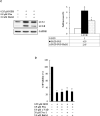
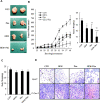
Methods: The anticancer effects of gemcitabine with pitavastatin were evaluated using human MIA PaCa-2 cell line in vitro and in vivo Balb/c murine xenograft tumor model. Cell viability was assessed with CCK-8, and cell migration was stained by crystal violet. Cell cycle distribution, apoptosis and mitochondrial membrane potential were examined by flow cytometry. Activation of drug transporters (hENTs, hCNTs), intracellular drug activating (dCK) and inhibition of inactivating enzymes (RRMs) pathways were assessed by Western blotting analysis. Molecular mechanisms and signaling pathways of apoptosis, necrosis and autophagy also were assessed by Western blotting.
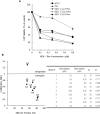
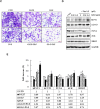
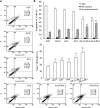
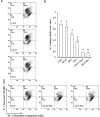
Results: We observed that gemcitabine and pitavastatin synergistically suppressed the proliferation of MIA PaCa-2 cells through causing sub-G1 and S phase cell cycle arrest. Activation of apoptosis/necrosis was confirmed by annexin V/propidium iodide double staining, which showed increasing levels of active caspase 3, cleaved poly(ADP-ribose) polymerase and the RIP1-RIP3-MLKL complex. Moreover, gemcitabine-pitavastatin-mediated S phase arrest downregulated cyclin A2/CDK2 and upregulated p21/p27 in MIA PaCa-2 cells. Furthermore, this combination improved drug cellular metabolism pathway, mitochondria function and activated autophagy as part of the cell death mechanism. In vivo, gemcitabine-pitavastatin effectively inhibited tumor growth in a nude mouse mode of Mia PaCa-2 xenografts without observed adverse effect.
Conclusion: Combined gemcitabine-pitavastatin may be an effective novel treatment option for pancreatic cancer.
Keywords: MIA PaCa-2; gemcitabine; pancreatic cancer; pitavastatin; synergistic anticancer.
© 2020 Chen et al.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures

Similar articles
-
Treatment with gemcitabine and TRA-8 anti-death receptor-5 mAb reduces pancreatic adenocarcinoma cell viability in vitro and growth in vivo.J Gastrointest Surg. 2006 Nov;10(9):1291-300; discussion 1300. doi: 10.1016/j.gassur.2006.08.007. J Gastrointest Surg. 2006. PMID: 17114015
-
Heptadecanoic Acid, an Odd-Chain Fatty Acid, Induces Apoptosis and Enhances Gemcitabine Chemosensitivity in Pancreatic Cancer Cells.J Med Food. 2023 Mar;26(3):201-210. doi: 10.1089/jmf.2022.K.0061. Epub 2023 Jan 30. J Med Food. 2023. PMID: 36716276
-
Enhanced Anti-tumor Effect of Flavopiridol in Combination With Gemcitabine in Pancreatic Cancer.Anticancer Res. 2024 Mar;44(3):1097-1108. doi: 10.21873/anticanres.16905. Anticancer Res. 2024. PMID: 38423644
-
Biodegradable nanocarrier of gemcitabine and tocopherol succinate synergistically ameliorates anti-proliferative response in MIA PaCa-2 cells.Int J Pharm. 2024 Jan 5;649:123599. doi: 10.1016/j.ijpharm.2023.123599. Epub 2023 Nov 21. Int J Pharm. 2024. PMID: 37992978
-
Pitavastatin and metformin synergistically activate apoptosis and autophagy in pancreatic cancer cells.Environ Toxicol. 2021 Aug;36(8):1491-1503. doi: 10.1002/tox.23146. Epub 2021 Apr 22. Environ Toxicol. 2021. PMID: 33886150
Cited by
-
Targeted Mevalonate Pathway and Autophagy in Antitumor Immunotherapy.Curr Cancer Drug Targets. 2024;24(9):890-909. doi: 10.2174/0115680096273730231206054104. Curr Cancer Drug Targets. 2024. PMID: 38275055 Review.
-
Mutant p53, the Mevalonate Pathway and the Tumor Microenvironment Regulate Tumor Response to Statin Therapy.Cancers (Basel). 2022 Jul 19;14(14):3500. doi: 10.3390/cancers14143500. Cancers (Basel). 2022. PMID: 35884561 Free PMC article. Review.
-
Critical Parameters to Improve Pancreatic Cancer Treatment Using Magnetic Hyperthermia: Field Conditions, Immune Response, and Particle Biodistribution.ACS Appl Mater Interfaces. 2021 Mar 24;13(11):12982-12996. doi: 10.1021/acsami.1c02338. Epub 2021 Mar 12. ACS Appl Mater Interfaces. 2021. PMID: 33709682 Free PMC article.
-
Exploring a repurposed candidate with dual hIDO1/hTDO2 inhibitory potential for anticancer efficacy identified through pharmacophore-based virtual screening and in vitro evaluation.Sci Rep. 2024 Apr 24;14(1):9386. doi: 10.1038/s41598-024-59353-4. Sci Rep. 2024. PMID: 38653790 Free PMC article.
-
Anti-tumor effect of statin on pancreatic adenocarcinoma: From concept to precision medicine.World J Clin Cases. 2021 Jun 26;9(18):4500-4505. doi: 10.12998/wjcc.v9.i18.4500. World J Clin Cases. 2021. PMID: 34222418 Free PMC article. Review.
References
-
- SEER Cancer Stat Facts: Pancreatic Cancer. Bethesda: National Cancer Institute; April 2019. Available from: https://seer.cancer.gov/statfacts/html/pancreas.html. Accessed May20, 2020.
-
- Labori KJ, Katz MH, Tzeng CW, et al. Impact of early disease progression and surgical complications on adjuvant chemotherapy completion rates and survival in patients undergoing the surgery first approach for resectable pancreatic ductal adenocarcinoma—a population-based cohort study. Acta Oncol. 2016;55:265–277. doi:10.3109/0284186X.2015.1068445 - DOI - PubMed
-
- Neoptolemos JP, Palmer DH, Ghaneh P, et al. European Study Group for Pancreatic Cancer. Comparison of adjuvant gemcitabine and capecitabine with gemcitabine monotherapy in patients with resected pancreatic cancer (ESPAC-4): a multicentre, open-label, randomised, Phase 3 trial. Lancet. 2017;389:1011–1024. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

