Assessment of the Release of Vascular Endothelial Growth Factor from 3D-Printed Poly-ε-Caprolactone/Hydroxyapatite/Calcium Sulfate Scaffold with Enhanced Osteogenic Capacity
- PMID: 32610580
- PMCID: PMC7408109
- DOI: 10.3390/polym12071455
Assessment of the Release of Vascular Endothelial Growth Factor from 3D-Printed Poly-ε-Caprolactone/Hydroxyapatite/Calcium Sulfate Scaffold with Enhanced Osteogenic Capacity
Abstract
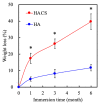
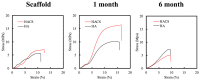
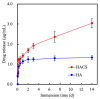
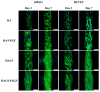
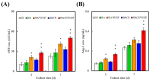
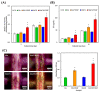
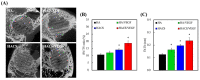
Vascular endothelial growth factor (VEGF) is one of the most crucial growth factors and an assistant for the adjustment of bone regeneration. In this study, a 3D scaffold is fabricated using the method of fused deposition modeling. Such a fabricated method allows us to fabricate scaffolds with consistent pore sizes, which could promote cellular ingrowth into scaffolds. Therefore, we drafted a plan to accelerate bone regeneration via VEGF released from the hydroxyapatite/calcium sulfate (HACS) scaffold. Herein, HACS will gradually degrade and provide a suitable environment for cell growth and differentiation. In addition, HACS scaffolds have higher mechanical properties and drug release compared with HA scaffolds. The drug release profile of the VEGF-loaded scaffolds showed that VEGF could be loaded and released in a stable manner. Furthermore, initial results showed that VEGF-loaded scaffolds could significantly enhance the proliferation of human mesenchymal stem cells (hMSCs) and human umbilical vein endothelial cells (HUVEC). In addition, angiogenic- and osteogenic-related proteins were substantially increased in the HACS/VEGF group. Moreover, in vivo results revealed that HACS/VEGF improved the regeneration of the rabbit's femur bone defect, and VEGF loading improved bone tissue regeneration and remineralization after implantation for 8 weeks. All these results strongly imply that the strategy of VEGF loading onto scaffolds could be a potential candidate for future bone tissue engineering.
Keywords: 3D printing; bone regeneration; calcium sulfate; hydroxyapatite; porous scaffold; vascular endothelial growth factor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
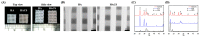



References
-
- Wang W.C., Nune K.C., Tan L., Zhang N., Dong J., Yan J., Misra RD K., Yang K. Bone regeneration of hollow tubular magnesium-strontium scaffolds in critical-size segmental defects: Effect of surface coatings. Mater. Sci. Eng. C Mater. Biol. Appl. 2019;100:297–307. doi: 10.1016/j.msec.2019.02.067. - DOI - PubMed
-
- He D., Dong W., Tang S., Wei J., Liu Z., Gu X., Li M., Guo H., Niu Y. Tissue engineering scaffolds of mesoporous magnesium silicate and poly(ε-caprolactone)-poly(ethylene glycol)-poly(ε-caprolactone) composite. J. Mater. Sci. Mater. Med. 2014;25:1415–1424. doi: 10.1007/s10856-014-5183-7. - DOI - PubMed
LinkOut - more resources
Full Text Sources

