Changes in relative histone abundance and heterochromatin in αA-crystallin and αB-crystallin knock-in mutant mouse lenses
- PMID: 32616056
- PMCID: PMC7331185
- DOI: 10.1186/s13104-020-05154-7
Changes in relative histone abundance and heterochromatin in αA-crystallin and αB-crystallin knock-in mutant mouse lenses
Abstract
Objective: Understanding the mechanisms of cataract formation is important for age-related and hereditary cataracts caused by mutations in lens protein genes. Lens proteins of the crystallin gene families α-, β-, and γ-crystallin are the most abundant proteins in the lens. Single point mutations in crystallin genes cause autosomal dominant cataracts in multigenerational families. Our previous proteomic and RNAseq studies identified genes and proteins altered in the early stages of cataract formation in mouse models. Histones H2A, H2B, and H4 increase in abundance in αA- and αB-crystallin mutant mouse lenses and in cultured cells expressing the mutant form of αA-crystallin linked with hereditary cataracts.
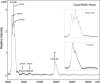
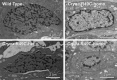
Results: In this study of histones in mutant lenses, we extracted histones from adult mouse lenses from cryaa-R49C and cryab-R120G mutant knock-in mice. We characterized the histones using matrix-assisted laser desorption/ionization time of flight (MALDI-TOF)-mass spectrometric analysis and gel electrophoresis and characterized the lens nucleus morphology using electron microscopy (EM). The relative abundance of histone H3 protein decreased in lenses from cryaa-R49C mutant mice and the relative abundance of histone H2 increased in these lenses. Electron microscopy of nuclei from cryaa-R49C-homozygous mutant mouse lenses revealed a pronounced alteration in the distribution of heterochromatin.
Keywords: Cataract; Crystallin; Electron microscopy; Histones; Mass spec; Mouse lens.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Probing the changes in gene expression due to α-crystallin mutations in mouse models of hereditary human cataract.PLoS One. 2018 Jan 16;13(1):e0190817. doi: 10.1371/journal.pone.0190817. eCollection 2018. PLoS One. 2018. PMID: 29338044 Free PMC article.
-
In vivo substrates of the lens molecular chaperones αA-crystallin and αB-crystallin.PLoS One. 2014 Apr 23;9(4):e95507. doi: 10.1371/journal.pone.0095507. eCollection 2014. PLoS One. 2014. PMID: 24760011 Free PMC article.
-
Autophagy and UPR in alpha-crystallin mutant knock-in mouse models of hereditary cataracts.Biochim Biophys Acta. 2016 Jan;1860(1 Pt B):234-9. doi: 10.1016/j.bbagen.2015.06.001. Epub 2015 Jun 11. Biochim Biophys Acta. 2016. PMID: 26071686 Free PMC article.
-
Effects of alpha-crystallin on lens cell function and cataract pathology.Curr Mol Med. 2009 Sep;9(7):887-92. doi: 10.2174/156652409789105598. Curr Mol Med. 2009. PMID: 19860667 Review.
-
Contrast Functions of αA- and αB-Crystallins in Cancer Development.Curr Mol Med. 2017;16(10):914-922. doi: 10.2174/1566524016666161223110508. Curr Mol Med. 2017. PMID: 28017134 Review.
Cited by
-
Changes in Protein Expression in Warmed Human Lens Epithelium Cells Using Shotgun Proteomics.Medicina (Kaunas). 2025 Feb 7;61(2):286. doi: 10.3390/medicina61020286. Medicina (Kaunas). 2025. PMID: 40005403 Free PMC article.
-
Zebrafish as a model for crystallin-associated congenital cataracts in humans.Front Cell Dev Biol. 2025 Mar 26;13:1552988. doi: 10.3389/fcell.2025.1552988. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40206405 Free PMC article. Review.
-
Single-Cell RNA Sequencing Analysis of the Early Postnatal Mouse Lens Epithelium.Invest Ophthalmol Vis Sci. 2023 Oct 3;64(13):37. doi: 10.1167/iovs.64.13.37. Invest Ophthalmol Vis Sci. 2023. PMID: 37870847 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

