Pyrrocidine, a molecular off switch for fumonisin biosynthesis
- PMID: 32628727
- PMCID: PMC7377494
- DOI: 10.1371/journal.ppat.1008595
Pyrrocidine, a molecular off switch for fumonisin biosynthesis
Abstract
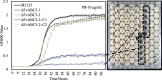
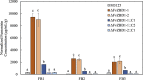
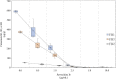
Sarocladium zeae is a fungal endophyte of maize and can be found co-inhabiting a single seed with Fusarium verticillioides, a major mycotoxigenic food safety threat. S. zeae produces pyrrocidines A and B that inhibit the growth of F. verticillioides and may limit its spread within the seed to locations lacking S. zeae. Although coinhabiting single seeds, the fungi are generally segregated in separate tissues. To understand F. verticillioides' protective physiological response to pyrrocidines we sequenced the F. verticillioides transcriptome upon exposure to purified pyrrocidine A or B at sub-inhibitory concentrations. Through this work we identified a F. verticillioides locus FvABC3 (FVEG_11089) encoding a transporter critical for resistance to pyrrocidine. We also identified FvZBD1 (FVEG_00314), a gene directly adjacent to the fumonisin biosynthetic gene cluster that was induced several thousand-fold in response to pyrrocidines. FvZBD1 is postulated to act as a genetic repressor of fumonisin production since deletion of the gene resulted in orders of magnitude increase in fumonisin. Further, pyrrocidine acts, likely through FvZBD1, to shut off fumonisin biosynthesis. This suggests that S. zeae is able to hack the secondary metabolic program of a competitor fungus, perhaps as preemptive self-protection, in this case impacting a mycotoxin of central concern for food safety.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures






References
-
- Ross PF, Nelson PE, Richard JL, Osweiler GD, Rice LG, Plattner RD, et al. Production of fumonisins by Fusarium moniliforme and Fusarium proliferatum isolates associated with equine leukoencephalomalacia and a pulmonary-edema syndrome in swine. Appl Environ Microbiol. 1990;56: 3225–3226. - PMC - PubMed
-
- Marasas WF, Kellerman TS, Gelderblom WC, Coetzer J a, Thiel PG, van der Lugt JJ. Leukoencephalomalacia in a horse induced by fumonisin B1 isolated from Fusarium moniliforme. Onderstepoort J Vet Res. 1988;55: 197–203. - PubMed
-
- King SB. Time of infection of maize kernels by Fusarium moniliforme and Cephalosporium acremonium. Phytopathology. 1981;71: 796 10.1094/Phyto-71-796 - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

