The Nuclear Lamina: Protein Accumulation and Disease
- PMID: 32630170
- PMCID: PMC7400325
- DOI: 10.3390/biomedicines8070188
The Nuclear Lamina: Protein Accumulation and Disease
Abstract
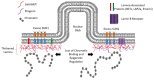
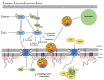
Cellular health is reliant on proteostasis-the maintenance of protein levels regulated through multiple pathways modulating protein synthesis, degradation and clearance. Loss of proteostasis results in serious disease and is associated with aging. One proteinaceous structure underlying the nuclear envelope-the nuclear lamina-coordinates essential processes including DNA repair, genome organization and epigenetic and transcriptional regulation. Loss of proteostasis within the nuclear lamina results in the accumulation of proteins, disrupting these essential functions, either via direct interactions of protein aggregates within the lamina or by altering systems that maintain lamina structure. Here we discuss the links between proteostasis and disease of the nuclear lamina, as well as how manipulating specific proteostatic pathways involved in protein clearance could improve cellular health and prevent/reverse disease.
Keywords: autophagy; clearance; lamina; neurodegeneration; premature aging; protein accumulation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
Publication types
LinkOut - more resources
Full Text Sources

