Light sheet microscopy of the gerbil cochlea
- PMID: 32632959
- PMCID: PMC7775904
- DOI: 10.1002/cne.24977
Light sheet microscopy of the gerbil cochlea
Abstract
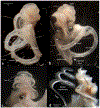
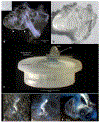
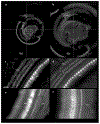
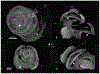
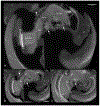
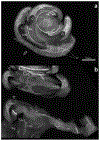
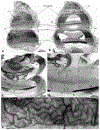
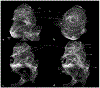
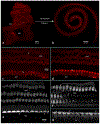
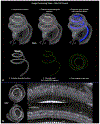
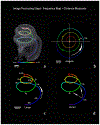
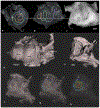
Light sheet fluorescence microscopy (LSFM) provides a rapid and complete three-dimensional image of the cochlea. The method retains anatomical relationships-on a micrometer scale-between internal structures such as hair cells, basilar membrane (BM), and modiolus with external surface structures such as the round and oval windows. Immunolabeled hair cells were used to visualize the spiraling BM in the intact cochlea without time intensive dissections or additional histological processing; yet material prepared for LSFM could be rehydrated, the BM dissected out and reimaged at higher resolution with the confocal microscope. In immersion-fixed material, details of the cochlear vasculature were seen throughout the cochlea. Hair cell counts (both inner and outer) as well as frequency maps of the BM were comparable to those obtained by other methods, but with the added dimension of depth. The material provided measures of angular, linear, and vector distance between characteristic frequency regions along the BM. Thus, LSFM provides a unique ability to rapidly image the entire cochlea in a manner applicable to model and interpret physiological results. Furthermore, the three-dimensional organization of the cochlea can be studied at the organ and cellular level with LSFM, and this same material can be taken to the confocal microscope for detailed analysis at the subcellular level.
Keywords: RRID: AB_10015251; RRID: AB_2534017; RRID:SCR_002465; RRID:SCR_003070; RRID:SCR_007370; auditory nerve; confocal microscopy; electrocochleography; inner hair cells; outer hair cells.
© 2020 Wiley Periodicals LLC.
Conflict of interest statement
Figures






References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

