A novel humanized mouse model to study the function of human cutaneous memory T cells in vivo in human skin
- PMID: 32636404
- PMCID: PMC7341892
- DOI: 10.1038/s41598-020-67430-7
A novel humanized mouse model to study the function of human cutaneous memory T cells in vivo in human skin
Abstract
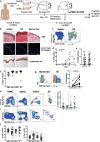
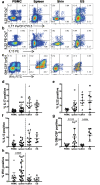
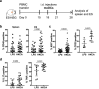
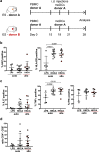
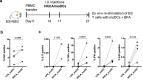
Human skin contains a population of memory T cells that supports tissue homeostasis and provides protective immunity. The study of human memory T cells is often restricted to in vitro studies and to human PBMC serving as primary cell source. Because the tissue environment impacts the phenotype and function of memory T cells, it is crucial to study these cells within their tissue. Here we utilized immunodeficient NOD-scid IL2rγnull (NSG) mice that carried in vivo-generated engineered human skin (ES). ES was generated from human keratinocytes and fibroblasts and was initially devoid of skin-resident immune cells. Upon adoptive transfer of human PBMC, this reductionist system allowed us to study human T cell recruitment from a circulating pool of T cells into non-inflamed human skin in vivo. Circulating human memory T cells preferentially infiltrated ES and showed diverse functional profiles of T cells found in fresh human skin. The chemokine and cytokine microenvironment of ES closely resembled that of non-inflamed human skin. Upon entering the ES T cells assumed a resident memory T cell-like phenotype in the absence of infection, and a proportion of these cutaneous T cells can be locally activated upon injection of monocyte derived dendritic cells (moDCs) that presented Candida albicans. Interestingly, we found that CD69+ memory T cells produced higher levels of effector cytokines in response to Candida albicans, compared to CD69- T cells. Overall, this model has broad utility in many areas of human skin immunology research, including the study of immune-mediated skin diseases.
Conflict of interest statement
M.M.K., E.M.M. and I.K.G. are inventors on patent application EP18168258 / US 16389821 that was jointly filed by the University of Salzburg, Austria, and Debra Austria that covers the use of skin humanized mice in pre-clinical studies involving engineered skin and human T cells.
Figures


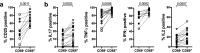
Similar articles
-
Staged development of long-lived T-cell receptor αβ TH17 resident memory T-cell population to Candida albicans after skin infection.J Allergy Clin Immunol. 2018 Aug;142(2):647-662. doi: 10.1016/j.jaci.2017.09.042. Epub 2017 Nov 9. J Allergy Clin Immunol. 2018. PMID: 29128674 Free PMC article.
-
Human skin is protected by four functionally and phenotypically discrete populations of resident and recirculating memory T cells.Sci Transl Med. 2015 Mar 18;7(279):279ra39. doi: 10.1126/scitranslmed.3010302. Sci Transl Med. 2015. PMID: 25787765 Free PMC article.
-
Long-term engraftment and expansion of tumor-derived memory T cells following the implantation of non-disrupted pieces of human lung tumor into NOD-scid IL2Rgamma(null) mice.J Immunol. 2008 May 15;180(10):7009-18. doi: 10.4049/jimmunol.180.10.7009. J Immunol. 2008. PMID: 18453623
-
Taking the lead - how keratinocytes orchestrate skin T cell immunity.Immunol Lett. 2018 Aug;200:43-51. doi: 10.1016/j.imlet.2018.06.009. Epub 2018 Jun 30. Immunol Lett. 2018. PMID: 29969603 Free PMC article. Review.
-
Human endothelial cell presentation of antigen and the homing of memory/effector T cells to skin.Ann N Y Acad Sci. 2001 Sep;941:12-25. doi: 10.1111/j.1749-6632.2001.tb03706.x. Ann N Y Acad Sci. 2001. PMID: 11594565 Review.
Cited by
-
The intestinal flora of patients with GHPA affects the growth and the expression of PD-L1 of tumor.Cancer Immunol Immunother. 2022 May;71(5):1233-1245. doi: 10.1007/s00262-021-03080-6. Epub 2021 Oct 13. Cancer Immunol Immunother. 2022. PMID: 34647152 Free PMC article.
-
Can Humanized Immune System Mouse and Rat Models Accelerate the Development of Cytomegalovirus-Based Vaccines Against Infectious Diseases and Cancers?Int J Mol Sci. 2025 Mar 27;26(7):3082. doi: 10.3390/ijms26073082. Int J Mol Sci. 2025. PMID: 40243710 Free PMC article. Review.
-
CD27-Expressing Xenoantigen-Expanded Human Regulatory T Cells Are Efficient in Suppressing Xenogeneic Immune Response.Cell Transplant. 2023 Jan-Dec;32:9636897221149444. doi: 10.1177/09636897221149444. Cell Transplant. 2023. PMID: 36644879 Free PMC article.
-
Generation and characterization of hair-bearing skin organoids from human pluripotent stem cells.Nat Protoc. 2022 May;17(5):1266-1305. doi: 10.1038/s41596-022-00681-y. Epub 2022 Mar 23. Nat Protoc. 2022. PMID: 35322210 Free PMC article. Review.
-
Tissue-Resident Memory T Cells in Skin Diseases: A Systematic Review.Int J Mol Sci. 2021 Aug 20;22(16):9004. doi: 10.3390/ijms22169004. Int J Mol Sci. 2021. PMID: 34445713 Free PMC article.
References
-
- Bos JD, et al. Predominance of ‘memory’ T cells (CD4+, CDw29+) over ‘naive’ T cells (CD4+, CD45R+) in both normal and diseased human skin. Arch. Dermatol. Res. 1989;281:24–30. - PubMed
-
- Clark RA, et al. The vast majority of CLA+ T cells are resident in normal skin. J. Immunol. Baltim. Md. 2006;1950(176):4431–4439. - PubMed
-
- Di Meglio P, Perera GK, Nestle FO. The multitasking organ: recent insights into skin immune function. Immunity. 2011;35:857–869. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

