Efficacy and Safety of Upadacitinib Monotherapy in Methotrexate-Naive Patients With Moderately-to-Severely Active Rheumatoid Arthritis (SELECT-EARLY): A Multicenter, Multi-Country, Randomized, Double-Blind, Active Comparator-Controlled Trial
- PMID: 32638504
- PMCID: PMC7589375
- DOI: 10.1002/art.41384
Efficacy and Safety of Upadacitinib Monotherapy in Methotrexate-Naive Patients With Moderately-to-Severely Active Rheumatoid Arthritis (SELECT-EARLY): A Multicenter, Multi-Country, Randomized, Double-Blind, Active Comparator-Controlled Trial
Abstract
Objective: The SELECT-EARLY trial was undertaken to study the effect of upadacitinib, an oral, reversible Janus kinase 1-selective inhibitor, as monotherapy in patients with predominantly early rheumatoid arthritis who were naive for or had limited exposure to methotrexate (MTX).
Methods: Patients (n = 947) were randomized 1:1:1 to receive once-daily doses of upadacitinib 15 mg or 30 mg or weekly MTX (7.5-20 mg/week) for 24 weeks. The primary end points were the proportion of patients who met the American College of Rheumatology 50% (ACR50) improvement criteria at week 12, and the proportion in whom a Disease Activity Score in 28 joints using the C-reactive protein level (DAS28-CRP) of <2.6 was achieved at week 24. Data are presented through week 24.
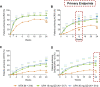
Results: At baseline, the median disease duration was 0.5 years (range 0-44 years). A total of 840 patients (89%) completed 24 weeks of treatment. The study met both primary end points for upadacitinib 15 mg and 30 mg versus MTX (ACR50 was achieved at week 12 in 52% and 56% of patients, respectively, versus 28% [P < 0.001], and DAS28-CRP <2.6 was achieved at week 24 in 48% and 50% of patients, respectively, versus 19% [P < 0.001]). Statistically significant and clinically meaningful improvements in multiple patient-reported outcomes (PROs) were recorded for both upadacitinib doses versus MTX. Overall, 88% of patients receiving upadacitinib 15 mg and 89% of patients receiving 30 mg, respectively, had no radiographic progression (modified total Sharp score ≤0) compared to 78% of those receiving MTX (P < 0.01). Through week 24, the frequency of treatment-emergent adverse events was similar between the MTX arm (65%) and upadacitinib 15 mg arm (64%), but was slightly higher in the upadacitinib 30 mg arm (71%). Six deaths were reported (2 in the upadacitinib 15 mg arm, 3 in the upadacitinib 30 mg arm, and 1 in the MTX arm).
Conclusion: Our findings indicate that patients receiving either dose of upadacitinib monotherapy experienced significant improvements in clinical, radiographic, and PROs compared to patients receiving MTX.
Trial registration: ClinicalTrials.gov NCT02706873.
© 2020 The Authors. Arthritis & Rheumatology published by Wiley Periodicals LLC on behalf of American College of Rheumatology.
Figures


Comment in
-
Upadacitinib in MTX-naive RA.Nat Rev Rheumatol. 2020 Sep;16(9):474. doi: 10.1038/s41584-020-0479-0. Nat Rev Rheumatol. 2020. PMID: 32699269 No abstract available.
References
-
- Singh JA, Saag KG, Bridges SL Jr, Akl EA, Bannuru RR, Sullivan MC, et al. 2015 American College of Rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Rheumatol 2016;68:1–26. - PubMed
-
- Smolen JS, Landewe R, Bijlsma J, Burmester G, Chatzidionysiou K, Dougados M, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease‐modifying antirheumatic drugs: 2016 update. Ann Rheum Dis 2017;76:960–77. - PubMed
-
- Emery P, Breedveld FC, Hall S, Durez P, Chang DJ, Robertson D, et al. Comparison of methotrexate monotherapy with a combination of methotrexate and etanercept in active, early, moderate to severe rheumatoid arthritis (COMET): a randomised, double‐blind, parallel treatment trial. Lancet 2008;372:375–82. - PubMed
-
- Van der Kooij SM, de Vries‐Bouwstra JK, Goekoop‐Ruiterman YP, van Zeben D, Kerstens PJ, Gerards AH, et al. Limited efficacy of conventional DMARDs after initial methotrexate failure in patients with recent onset rheumatoid arthritis treated according to the disease activity score. Ann Rheum Dis 2007;66:1356–62. - PMC - PubMed
-
- Van Vollenhoven RF, Ernestam S, Geborek P, Petersson IF, Cöster L, Waltbrand E, et al. Addition of infliximab compared with addition of sulfasalazine and hydroxychloroquine to methotrexate in patients with early rheumatoid arthritis (SWEFOT trial): 1‐year results of a randomised trial. Lancet 2009;374:459–66. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

