Microfluidic enrichment of bacteria coupled to contact-free lysis on a magnetic polymer surface for downstream molecular detection
- PMID: 32642021
- PMCID: PMC7316515
- DOI: 10.1063/5.0011908
Microfluidic enrichment of bacteria coupled to contact-free lysis on a magnetic polymer surface for downstream molecular detection
Abstract
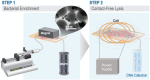
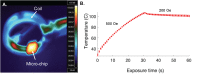
We report on a microsystem that couples high-throughput bacterial immunomagnetic capture to contact-free cell lysis using an alternating current magnetic field (AMF) to enable downstream molecular characterization of bacterial nucleic acids. Traditional methods for cell lysis rely on either dilutive chemical methods, expensive biological reagents, or imprecise physical methods. We present a microchip with a magnetic polymer substrate (Mag-Polymer microchip), which enables highly controlled, on-chip heating of biological targets following exposure to an AMF. First, we present a theoretical framework for the quantitation of power generation for single-domain magnetic nanoparticles embedded in a polymer matrix. Next, we demonstrate successful bacterial DNA recovery by coupling (1) high-throughput, sensitive microfluidic immunomagnetic capture of bacteria to (2) on-chip, contact-free bacterial lysis using an AMF. The bacterial capture efficiency exceeded 76% at 50 ml/h at cell loads as low as ∼10 CFU/ml, and intact DNA was successfully recovered at starting bacterial concentrations as low as ∼1000 CFU/ml. Using the presented methodology, cell lysis becomes non-dilutive, temperature is precisely controlled, and potential contamination risks are eliminated. This workflow and substrate modification could be easily integrated in a range of micro-scale diagnostic systems for infectious disease.
Copyright © 2020 Author(s).
Figures


Similar articles
-
Polymer-based microfluidic chip for rapid and efficient immunomagnetic capture and release of Listeria monocytogenes.Lab Chip. 2015 Oct 21;15(20):3994-4007. doi: 10.1039/c5lc00852b. Epub 2015 Sep 8. Lab Chip. 2015. PMID: 26346021
-
Lysis of gram-positive and gram-negative bacteria by antibacterial porous polymeric monolith formed in microfluidic biochips for sample preparation.Anal Bioanal Chem. 2014 Sep;406(24):5977-87. doi: 10.1007/s00216-014-8028-9. Epub 2014 Jul 25. Anal Bioanal Chem. 2014. PMID: 25059724
-
Performance Evaluation of Fast Microfluidic Thermal Lysis of Bacteria for Diagnostic Sample Preparation.Diagnostics (Basel). 2013 Jan 17;3(1):105-16. doi: 10.3390/diagnostics3010105. Diagnostics (Basel). 2013. PMID: 26835670 Free PMC article.
-
Magnetic particles for in vitro molecular diagnosis: From sample preparation to integration into microsystems.Colloids Surf B Biointerfaces. 2017 Oct 1;158:1-8. doi: 10.1016/j.colsurfb.2017.06.024. Epub 2017 Jun 22. Colloids Surf B Biointerfaces. 2017. PMID: 28654866 Review.
-
Integrated microfluidic systems for DNA analysis.Top Curr Chem. 2011;304:203-60. doi: 10.1007/128_2011_153. Top Curr Chem. 2011. PMID: 21607848 Review.
Cited by
-
Leapfrogging laboratories: the promise and pitfalls of high-tech solutions for antimicrobial resistance surveillance in low-income settings.BMJ Glob Health. 2020 Dec;5(12):e003622. doi: 10.1136/bmjgh-2020-003622. BMJ Glob Health. 2020. PMID: 33268385 Free PMC article. Review.
-
Review of Microfluidic Methods for Cellular Lysis.Micromachines (Basel). 2021 Apr 28;12(5):498. doi: 10.3390/mi12050498. Micromachines (Basel). 2021. PMID: 33925101 Free PMC article. Review.
-
Magnetic nanoparticles fabricated/integrated with microfluidics for biological applications: A review.Biomed Microdevices. 2024 Jan 25;26(1):13. doi: 10.1007/s10544-023-00693-9. Biomed Microdevices. 2024. PMID: 38270676 Review.
-
Advances in the use of nanomaterials for nucleic acid detection in point-of-care testing devices: A review.Front Bioeng Biotechnol. 2022 Oct 13;10:1020444. doi: 10.3389/fbioe.2022.1020444. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36312553 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
