Hyperhomocysteinemia and Low Folate and Vitamin B12 Are Associated with Vascular Dysfunction and Impaired Nitric Oxide Sensitivity in Morbidly Obese Patients
- PMID: 32645905
- PMCID: PMC7400872
- DOI: 10.3390/nu12072014
Hyperhomocysteinemia and Low Folate and Vitamin B12 Are Associated with Vascular Dysfunction and Impaired Nitric Oxide Sensitivity in Morbidly Obese Patients
Abstract
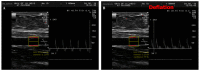
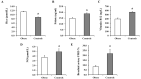
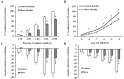
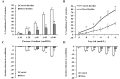
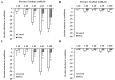
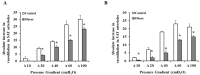
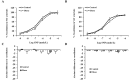
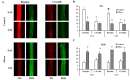
There is a high prevalence of hyperhomocysteinemia that has been linked to high cardiovascular risk in obese individuals and could be attributed to poor nutritional status of folate and vitamin B12. We sought to examine the association between blood homocysteine (Hcy) folate, and vitamin B12 levels and vascular dysfunction in morbidly obese adults using novel ex vivo flow-induced dilation (FID) measurements of isolated adipose tissue arterioles. Brachial artery flow-mediated dilation (FMD) was also measured. Subcutaneous and visceral adipose tissue biopsies were obtained from morbidly obese individuals and non-obese controls. Resistance arterioles were isolated in which FID, acetylcholine-induced dilation (AChID), and nitric oxide (NO) production were measured in the absence or presence of the NO synthase inhibitor, L-NAME, Hcy, or the superoxide dismutase mimetic, TEMPOL. Our results demonstrated that plasma Hcy concentrations were significantly higher, while folate, vitamin B12, and NO were significantly lower in obese subjects compared to controls. Hcy concentrations correlated positively with BMI, fat %, and insulin levels but not with folate or vitamin B12. Brachial and arteriolar vasodilation were lower in obese subjects, positively correlated with folate and vitamin B12, and inversely correlated with Hcy. Arteriolar NO measurements and sensitivity to L-NAME were lower in obese subjects compared to controls. Finally, Hcy incubation reduced arteriolar FID and NO sensitivity, an effect that was abolished by TEMPOL. In conclusion, these data suggest that high concentrations of plasma Hcy and low concentrations of folate and vitamin B12 could be independent predictors of vascular dysfunction in morbidly obese individuals.
Keywords: bariatric surgery; folate; homocysteine; nitric oxide; obesity; vascular dysfunction; vitamin B12.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Association of B vitamins status and homocysteine levels in elderly Taiwanese.Asia Pac J Clin Nutr. 2005;14(3):250-5. Asia Pac J Clin Nutr. 2005. PMID: 16169836
-
Vitamin B12 deficiency and hyperhomocysteinemia as correlates of cardiovascular risk factors in Indian subjects with coronary artery disease.J Cardiol. 2013 Apr;61(4):289-94. doi: 10.1016/j.jjcc.2012.11.009. Epub 2013 Mar 6. J Cardiol. 2013. PMID: 23473764
-
Total plasma homocysteine, folate, and vitamin B12 status in healthy Iranian adults: the Tehran homocysteine survey (2003-2004)/a cross-sectional population based study.BMC Public Health. 2006 Feb 13;6:29. doi: 10.1186/1471-2458-6-29. BMC Public Health. 2006. PMID: 16472406 Free PMC article.
-
Vitamin B12, folate, and homocysteine levels and multiple sclerosis: A meta-analysis.Mult Scler Relat Disord. 2017 Oct;17:190-197. doi: 10.1016/j.msard.2017.08.004. Epub 2017 Aug 16. Mult Scler Relat Disord. 2017. PMID: 29055456 Review.
-
Indicators for assessing folate and vitamin B12 status and for monitoring the efficacy of intervention strategies.Food Nutr Bull. 2008 Jun;29(2 Suppl):S52-63; discussion S64-6. doi: 10.1177/15648265080292S108. Food Nutr Bull. 2008. PMID: 18709881 Review.
Cited by
-
DNA methylation profile of genes involved in inflammation and autoimmunity correlates with vascular function in morbidly obese adults.Epigenetics. 2022 Jan;17(1):93-109. doi: 10.1080/15592294.2021.1876285. Epub 2021 Jan 25. Epigenetics. 2022. PMID: 33487124 Free PMC article.
-
Obesity-Associated Vitamin D Deficiency Correlates with Adipose Tissue DNA Hypomethylation, Inflammation, and Vascular Dysfunction.Int J Mol Sci. 2022 Nov 19;23(22):14377. doi: 10.3390/ijms232214377. Int J Mol Sci. 2022. PMID: 36430854 Free PMC article.
-
Time to Change Our Viewpoints to Assess Renal Risks in Patients with Solitary Kidneys beyond Traditional Approaches?J Clin Med. 2023 Oct 31;12(21):6885. doi: 10.3390/jcm12216885. J Clin Med. 2023. PMID: 37959350 Free PMC article.
-
The damaging duo: Obesity and excess dietary salt contribute to hypertension and cardiovascular disease.Obes Rev. 2023 Aug;24(8):e13589. doi: 10.1111/obr.13589. Epub 2023 Jun 19. Obes Rev. 2023. PMID: 37336641 Free PMC article. Review.
-
The carotenoid torularhodin alleviates NAFLD by promoting Akkermanisa muniniphila-mediated adenosylcobalamin metabolism.Nat Commun. 2025 Apr 8;16(1):3338. doi: 10.1038/s41467-025-58500-3. Nat Commun. 2025. PMID: 40199868 Free PMC article.
References
-
- Poli V.F.S., Sanches R.B., Moraes A.D.S., Fidalgo J.P.N., Nascimento M.A., Bresciani P., Andrade-Silva S.G., Cipullo M.A.T., Clemente J.C., Caranti D.A. The excessive caloric intake and micronutrient deficiencies related to obesity after a long-term interdisciplinary therapy. Nutrition. 2017;38:113–119. doi: 10.1016/j.nut.2017.01.012. - DOI - PubMed
-
- Selhub J. Folate, vitamin B12 and vitamin B6 and one carbon metabolism. J. Nutr. Health Aging. 2002;6:39–42. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

