Programmable Nanodisc Patterning by DNA Origami
- PMID: 32648765
- PMCID: PMC7814775
- DOI: 10.1021/acs.nanolett.0c02048
Programmable Nanodisc Patterning by DNA Origami
Abstract
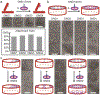
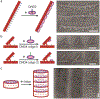
Nanodiscs (ND) are soluble phospholipid bilayers bounded by membrane scaffold proteins; they have become invaluable in the study of membrane proteins. However, this multifunctional tool has been used individually, and applications involving multiple NDs and their interactions have fallen far behind their counterpart membrane model system: liposomes. One major obstacle is the lack of reliable methods to manage the spatial arrangement of NDs. Here we sought to extend the utility of NDs by organizing them on DNA origami. NDs constructed with DNA-anchor amphiphiles were placed precisely and specifically into these DNA nanostructures via hybridization. Four different tethering strategies were explored and validated. A variety of geometric patterns of NDs were successfully programmed on origami, as evidenced by electron microscopy. The ND ensembles generated in this study provide new and powerful platforms to study protein-lipid or protein-protein interactions with spatial control of membranes.
Keywords: DNA origami; DNA−protein conjugation; membrane proteins; nanodisc.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
DNA-nanostructure-templated assembly of planar and curved lipid-bilayer membranes.Front Chem. 2023 Feb 8;10:1047874. doi: 10.3389/fchem.2022.1047874. eCollection 2022. Front Chem. 2023. PMID: 36844038 Free PMC article. Review.
-
The nanodisc: a novel tool for membrane protein studies.Biol Chem. 2009 Aug;390(8):805-14. doi: 10.1515/BC.2009.091. Biol Chem. 2009. PMID: 19453280 Review.
-
Conformational transitions in the membrane scaffold protein of phospholipid bilayer nanodiscs.Mol Cell Proteomics. 2011 Sep;10(9):M111.010876. doi: 10.1074/mcp.M111.010876. Epub 2011 Jun 29. Mol Cell Proteomics. 2011. PMID: 21715319 Free PMC article.
-
Properties of DNA- and Protein-Scaffolded Lipid Nanodiscs.ACS Nano. 2021 Jan 26;15(1):751-764. doi: 10.1021/acsnano.0c07128. Epub 2020 Dec 21. ACS Nano. 2021. PMID: 33347276
-
A programmable DNA-origami platform for studying lipid transfer between bilayers.Nat Chem Biol. 2019 Aug;15(8):830-837. doi: 10.1038/s41589-019-0325-3. Epub 2019 Jul 18. Nat Chem Biol. 2019. PMID: 31320758 Free PMC article.
Cited by
-
Digital nanoreactors to control absolute stoichiometry and spatiotemporal behavior of DNA receptors within lipid bilayers.Nat Commun. 2023 Mar 20;14(1):1532. doi: 10.1038/s41467-023-36996-x. Nat Commun. 2023. PMID: 36941256 Free PMC article.
-
One-step construction of circularized nanodiscs using SpyCatcher-SpyTag.Nat Commun. 2021 Sep 14;12(1):5451. doi: 10.1038/s41467-021-25737-7. Nat Commun. 2021. PMID: 34521837 Free PMC article.
-
Functionalizing DNA origami to investigate and interact with biological systems.Nat Rev Mater. 2023 Feb;8(2):123-138. doi: 10.1038/s41578-022-00517-x. Epub 2022 Dec 19. Nat Rev Mater. 2023. PMID: 37206669 Free PMC article.
-
[Application of DNA origami in nanobiomedicine].Nan Fang Yi Ke Da Xue Xue Bao. 2021 Jun 20;41(6):960-964. doi: 10.12122/j.issn.1673-4254.2021.06.22. Nan Fang Yi Ke Da Xue Xue Bao. 2021. PMID: 34238752 Free PMC article. Review. Chinese.
-
Synaptotagmin 1 oligomerization via the juxtamembrane linker regulates spontaneous and evoked neurotransmitter release.Proc Natl Acad Sci U S A. 2021 Nov 30;118(48):e2113859118. doi: 10.1073/pnas.2113859118. Proc Natl Acad Sci U S A. 2021. PMID: 34810248 Free PMC article.
References
-
- Bayburt TH; Grinkova YV; Sligar SG Self-assembly of discoidal phospholipid bilayer nanoparticles with membrane scaffold proteins. Nano Lett. 2002, 2, 853–856.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

