Tract-specific analysis improves sensitivity of spinal cord diffusion MRI to cross-sectional and longitudinal changes in amyotrophic lateral sclerosis
- PMID: 32651439
- PMCID: PMC7351722
- DOI: 10.1038/s42003-020-1093-z
Tract-specific analysis improves sensitivity of spinal cord diffusion MRI to cross-sectional and longitudinal changes in amyotrophic lateral sclerosis
Abstract
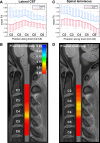
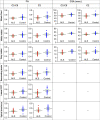
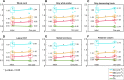
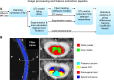
Amyotrophic lateral sclerosis (ALS) is a late-onset fatal neurodegenerative disease that causes progressive degeneration of motor neurons in the brain and the spinal cord. Corticospinal tract degeneration is a defining feature of ALS. However, there have been very few longitudinal, controlled studies assessing diffusion MRI (dMRI) metrics in different fiber tracts along the spinal cord in general or the corticospinal tract in particular. Here we demonstrate that a tract-specific analysis, with segmentation of ascending and descending tracts in the spinal cord white matter, substantially increases the sensitivity of dMRI to disease-related changes in ALS. Our work also identifies the tracts and spinal levels affected in ALS, supporting electrophysiologic and pathologic evidence of involvement of sensory pathways in ALS. We note changes in diffusion metrics and cord cross-sectional area, with enhanced sensitivity to disease effects through a multimodal analysis, and with strong correlations between these metrics and spinal components of ALSFRS-R.
Conflict of interest statement
The authors declare no competing interests.
Figures




References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

