Interactions between Borrelia burgdorferi and ticks
- PMID: 32651470
- PMCID: PMC7351536
- DOI: 10.1038/s41579-020-0400-5
Interactions between Borrelia burgdorferi and ticks
Abstract
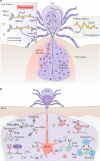
Borrelia burgdorferi is the causative agent of Lyme disease and is transmitted to vertebrate hosts by Ixodes spp. ticks. The spirochaete relies heavily on its arthropod host for basic metabolic functions and has developed complex interactions with ticks to successfully colonize, persist and, at the optimal time, exit the tick. For example, proteins shield spirochaetes from immune factors in the bloodmeal and facilitate the transition between vertebrate and arthropod environments. On infection, B. burgdorferi induces selected tick proteins that modulate the vector gut microbiota towards an environment that favours colonization by the spirochaete. Additionally, the recent sequencing of the Ixodes scapularis genome and characterization of tick immune defence pathways, such as the JAK-STAT, immune deficiency and cross-species interferon-γ pathways, have advanced our understanding of factors that are important for B. burgdorferi persistence in the tick. In this Review, we summarize interactions between B. burgdorferi and I. scapularis during infection, as well as interactions with tick gut and salivary gland proteins important for establishing infection and transmission to the vertebrate host.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
-
- Fraser CM, et al. Genomic sequence of a Lyme disease spirochaete, Borrelia burgdorferi. Nature. 1997;390:580–586. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

