Regulation of V-ATPase Activity and Organelle pH by Phosphatidylinositol Phosphate Lipids
- PMID: 32656214
- PMCID: PMC7324685
- DOI: 10.3389/fcell.2020.00510
Regulation of V-ATPase Activity and Organelle pH by Phosphatidylinositol Phosphate Lipids
Abstract
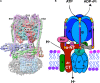
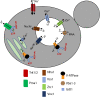
Luminal pH and the distinctive distribution of phosphatidylinositol phosphate (PIP) lipids are central identifying features of organelles in all eukaryotic cells that are also critical for organelle function. V-ATPases are conserved proton pumps that populate and acidify multiple organelles of the secretory and the endocytic pathway. Complete loss of V-ATPase activity causes embryonic lethality in higher animals and conditional lethality in yeast, while partial loss of V-ATPase function is associated with multiple disease states. On the other hand, many cancer cells increase their virulence by upregulating V-ATPase expression and activity. The pH of individual organelles is tightly controlled and essential for function, but the mechanisms for compartment-specific pH regulation are not completely understood. There is substantial evidence indicating that the PIP content of membranes influences organelle pH. We present recent evidence that PIPs interact directly with subunit isoforms of the V-ATPase to dictate localization of V-ATPase subpopulations and participate in their regulation. In yeast cells, which have only one set of organelle-specific V-ATPase subunit isoforms, the Golgi-enriched lipid PI(4)P binds to the cytosolic domain of the Golgi-enriched a-subunit isoform Stv1, and loss of PI(4)P binding results in mislocalization of Stv1-containing V-ATPases from the Golgi to the vacuole/lysosome. In contrast, levels of the vacuole/lysosome-enriched signaling lipid PI(3,5)P2 affect assembly and activity of V-ATPases containing the Vph1 a-subunit isoform. Mutations in the Vph1 isoform that disrupt the lipid interaction increase sensitivity to stress. These studies have decoded "zip codes" for PIP lipids in the cytosolic N-terminal domain of the a-subunit isoforms of the yeast V-ATPase, and similar interactions between PIP lipids and the V-ATPase subunit isoforms are emerging in higher eukaryotes. In addition to direct effects on the V-ATPase, PIP lipids are also likely to affect organelle pH indirectly, through interactions with other membrane transporters. We discuss direct and indirect effects of PIP lipids on organelle pH, and the functional consequences of the interplay between PIP lipid content and organelle pH.
Keywords: Golgi apparatus; PIKfyve; V-ATPase; acidification; endosome; lysosome; organelle; phosphatidylinositol phosphate.
Copyright © 2020 Banerjee and Kane.
Figures


References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

