WNT Signaling in Melanoma
- PMID: 32659938
- PMCID: PMC7402324
- DOI: 10.3390/ijms21144852
WNT Signaling in Melanoma
Abstract
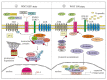
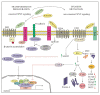
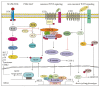
WNT-signaling controls important cellular processes throughout embryonic development and adult life, so any deregulation of this signaling can result in a wide range of pathologies, including cancer. WNT-signaling is classified into two categories: β-catenin-dependent signaling (canonical pathway) and β-catenin-independent signaling (non-canonical pathway), the latter can be further divided into WNT/planar cell polarity (PCP) and calcium pathways. WNT ligands are considered as unique directional growth factors that contribute to both cell proliferation and polarity. Origin of cancer can be diverse and therefore tissue-specific differences can be found in WNT-signaling between cancers, including specific mutations contributing to cancer development. This review focuses on the role of the WNT-signaling pathway in melanoma. The current view on the role of WNT-signaling in cancer immunity as well as a short summary of WNT pathway-related drugs under investigation are also provided.
Keywords: WNT; WNT5A; immune evasion; melanoma; signal transduction crosstalk; β-catenin.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

