A Multimethod, Multicountry Evaluation of Breakpoints for Bedaquiline Resistance Determination
- PMID: 32660992
- PMCID: PMC7449194
- DOI: 10.1128/AAC.00479-20
A Multimethod, Multicountry Evaluation of Breakpoints for Bedaquiline Resistance Determination
Abstract
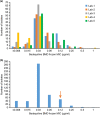
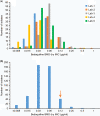
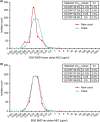
Criteria defining bedaquiline resistance for tuberculosis have been proposed addressing an emerging concern. We evaluated bedaquiline phenotypic drug susceptibility testing (pDST) criteria using drug-resistant tuberculosis clinical isolates tested at five reference laboratories. Isolates were tested at the proposed bedaquiline MGIT960 and 7H11 agar proportion (AP) critical concentrations and also at higher dilutions. The epidemiological cutoff value for the broth microdilution (BMD) plates (frozen and dry) was investigated. Sanger sequencing was performed (atpE and Rv0678 genes) for any isolate testing resistant. The composite reference standard (CRS) defined susceptibility or resistance as is if all pDST methods agreed. If the pDST result was discordant, sequencing results were used for final classification. Geographically diverse and bedaquiline-unexposed isolates were tested (n = 495). The epidemiological cutoff value for BMD was confirmed to be 0.12 μg/ml. The majority of isolates were determined to be susceptible by all methods (467/495; 94.3%), and 28 were determined to be resistant by at least one method; 4 of these were determined to be resistant by all methods. Of the 28 resistant isolates, 12 harbored Rv0678 mutations exclusively. Isolates with insertions/deletions were more likely to be determined to be resistant by more than one method (5/7) compared to isolates with a single nucleotide polymorphism (1/5). Applying the CRS to 24 discordant pDST, BMD dry correctly detected most (15/24; 63%), followed by MGIT960 and BMD frozen (13/24; 61%) and lastly AP (12/24; 50%). Applying the CRS, the prevalence of bedaquiline resistance was 2.2% and ranged from 1.4 to 3.4%, depending on the method used. All methods performed well for bedaquiline susceptibility determination; however, resistance detected should be investigated by a second, alternative method.
Keywords: Mycobacterium tuberculosis; bedaquiline; drug resistance; drug susceptibility testing; tuberculosis.
Copyright © 2020 American Society for Microbiology.
Figures

References
-
- World Health Organization. Global TB Report. 2018. World Health Organization, Geneva, Switzerland: http://apps.who.int/iris/bitstream/handle/10665/274453/9789241565646-eng.... Accessed 7 December 2018.
-
- Andries K, Verhasselt P, Guillemont J, Göhlmann WH, Neefs J-M, Winkler H, Van Gestel J, Timmerman P, Zhu M, Lee E, Williams P, de Chaffoy D, Huitric E, Hoffner S, Cambau E, Truffot-Pernot C, Lounis N, Jarlier V. 2005. A diarylquinoline drug active on the ATP synthase of Mycobacterium tuberculosis. Science 307:223–227. doi:10.1126/science.1106753. - DOI - PubMed
-
- Schnippel K, Ndjeka N, Maartens G, Meintjes G, Master I, Ismail N, Hughes J, Ferreira H, Padanilam X, Romero R, Te Riele J, Conradie F. 2018. Effect of bedaquiline on mortality in South African patients with drug-resistant tuberculosis: a retrospective cohort study. Lancet Respir Med 6:699–706. doi:10.1016/S2213-2600(18)30235-2. - DOI - PubMed
-
- Mbuagbaw L, Guglielmetti L, Hewison C, Bakare N, Bastard M, Caumes E, Fréchet-Jachym M, Robert J, Veziris N, Khachatryan N, Kotrikadze T, Hayrapetyan A, Avaliani Z, Schünemann HJ, Lienhardt C. 2019. Outcomes of bedaquiline treatment in patients with multidrug-resistant tuberculosis. Emerg Infect Dis 25:936–943. doi:10.3201/eid2505.181823. - DOI - PMC - PubMed
-
- Diacon AH, Donald PR, Pym A, Grobusch M, Patientia RF, Mahanyele R, Bantubani N, Narasimooloo R, De Marez T, van Heeswijk R, Lounis N, Meyvisch P, Andries K, McNeeley DF. 2012. Randomized pilot trial of eight weeks of bedaquiline (TMC207) treatment for multidrug-resistant tuberculosis: long-term outcome, tolerability, and effect on emergence of drug resistance. Antimicrob Agents Chemother 56:3271–3276. doi:10.1128/AAC.06126-11. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

