Granulocyte Colony Stimulating Factor (GCSF) Can Attenuate Neuropathic Pain by Suppressing Monocyte Chemoattractant Protein-1 (MCP-1) Expression, through Upregulating the Early MicroRNA-122 Expression in the Dorsal Root Ganglia
- PMID: 32664488
- PMCID: PMC7408430
- DOI: 10.3390/cells9071669
Granulocyte Colony Stimulating Factor (GCSF) Can Attenuate Neuropathic Pain by Suppressing Monocyte Chemoattractant Protein-1 (MCP-1) Expression, through Upregulating the Early MicroRNA-122 Expression in the Dorsal Root Ganglia
Abstract
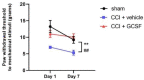
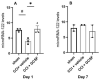
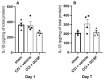
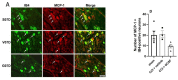
Our previous animal studies and several human clinical trials have shown that granulocyte-colony stimulating factor (GCSF) can attenuate neuropathic pain through various mechanisms. GCSF itself is also a multipotent cytokine that can modulate microribonucleic acid (microRNA) expression profiles in vitro. In this study, we used the NanoString nCounter analysis system to screen the expression of different rodent microRNAs at early stage after nerve injury and studied the expression of related cytokines/chemokines in the dorsal root ganglia (DRGs) of rats that underwent chronic constriction injury (CCI) to explore the underlying mechanisms of the analgesic effects of GCSF. We found that microRNA-122 expression was downregulated by CCI; in contrast, GCSF treatment significantly upregulated microRNA-122 expression in the DRGs of CCI rats on the 1st day after nerve injury. We further studied the expression of different cytokines/chemokines (IL-1β, IL-6, and monocyte chemoattractant protein-1 (MCP-1)) that were modulated by microRNA-122. MCP-1 has been reported to participate in neuropathic pain development, and its expression on the DRGs of vehicle-treated CCI rats was significantly higher than that on the DRGs of sham-operated rats; in contrast, GCSF-treated rats exhibited significantly lower MCP-1 expression in the DRG than vehicle-treated rats on the 7th day after nerve injury. An early GCSF treatment can suppress MCP-1 expressions, through upregulating microRNA-122 expressions in the DRGs of CCI rats at an earlier stage, thus indirectly attenuating neuropathic pain development.
Keywords: chronic constriction injury; dorsal root ganglia; granulocyte colony stimulating factor; microRNA-122; monocyte chemoattractant protein-1; neuropathic pain; rats.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Koda M., Furuya T., Kato K., Mannoji C., Hashimoto M., Inada T., Kamiya K., Ota M., Maki S., Okawa A., et al. Delayed granulocyte colony-stimulating factor treatment in rats attenuates mechanical allodynia induced by chronic constriction injury of the sciatic nerve. Spine. 2014;39:192–197. doi: 10.1097/BRS.0000000000000108. - DOI - PubMed
-
- Kato K., Koda M., Takahashi H., Sakuma T., Inada T., Kamiya K., Ota M., Maki S., Okawa A., Takahashi K., et al. Granulocyte colony-stimulating factor attenuates spinal cord injury-induced mechanical allodynia in adult rats. J. Neurol. Sci. 2015;355:79–83. doi: 10.1016/j.jns.2015.05.024. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

