Thrombospondin-1 Plays an Essential Role in Yes-Associated Protein Nuclear Translocation during the Early Phase of Trypanosoma cruzi Infection in Heart Endothelial Cells
- PMID: 32664627
- PMCID: PMC7403984
- DOI: 10.3390/ijms21144912
Thrombospondin-1 Plays an Essential Role in Yes-Associated Protein Nuclear Translocation during the Early Phase of Trypanosoma cruzi Infection in Heart Endothelial Cells
Abstract
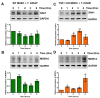
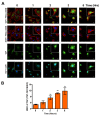
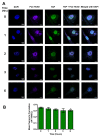
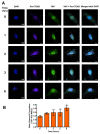
The protozoan parasite Trypanosoma cruzi is the causative agent of Chagas disease. This neglected tropical disease causes severe morbidity and mortality in endemic regions. About 30% of T. cruzi infected individuals will present with cardiac complications. Invasive trypomastigotes released from infected cells can be carried in the vascular endothelial system to infect neighboring and distant cells. During the process of cellular infection, the parasite induces host cells, to increase the levels of host thrombospondin-1 (TSP-1), to facilitate the process of infection. TSP-1 plays important roles in the functioning of vascular cells, including vascular endothelial cells with important implications in cardiovascular health. Many signal transduction pathways, including the yes-associated protein 1 (YAP)/transcriptional coactivator, with PDZ-binding motif (TAZ) signaling, which are upstream of TSP-1, have been linked to the pathophysiology of heart damage. The molecular mechanisms by which T. cruzi signals, and eventually infects, heart endothelial cells remain unknown. To evaluate the importance of TSP-1 expression in heart endothelial cells during the process of T. cruzi infection, we exposed heart endothelial cells prepared from Wild Type and TSP-1 Knockout mouse to invasive T. cruzi trypomastigotes at multiple time points, and evaluated changes in the hippo signaling cascade using immunoblotting and immunofluorescence assays. We found that the parasite turned off the hippo signaling pathway in TSP-1KO heart endothelial cells. The levels of SAV1 and MOB1A increased to a maximum of 2.70 ± 0.23 and 5.74 ± 1.45-fold at 3 and 6 h, respectively, in TSP-1KO mouse heart endothelial cells (MHEC), compared to WT MHEC, following a parasite challenge. This was accompanied by a significant continuous increase in the nuclear translocation of downstream effector molecule YAP, to a maximum mean nuclear fluorescence intensity of 10.14 ± 0.40 at 6 h, compared to wild type cells. Furthermore, we found that increased nuclear translocated YAP significantly colocalized with the transcription co-activator molecule pan-TEAD, with a maximum Pearson's correlation coefficient of 0.51 ± 0.06 at 6 h, compared to YAP-Pan-TEAD colocalization in the WT MHEC, which decreased significantly, with a minimum Pearson's correlation coefficient of 0.30 ± 0.01 at 6 h. Our data indicate that, during the early phase of infection, upregulated TSP-1 is essential for the regulation of the hippo signaling pathway. These studies advance our understanding of the molecular interactions occurring between heart endothelial cells and T. cruzi, in the presence and absence of TSP-1, providing insights into processes linked to parasite dissemination and pathogenesis.
Keywords: Chagas heart disease; Trypanosoma cruzi; heart endothelial cells; hippo signaling; transcriptional enhancer factor (TEF) family/TEA domain (TEAD) family; yes-associated protein1.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

