SARS-CoV-2 induces transcriptional signatures in human lung epithelial cells that promote lung fibrosis
- PMID: 32664949
- PMCID: PMC7359430
- DOI: 10.1186/s12931-020-01445-6
SARS-CoV-2 induces transcriptional signatures in human lung epithelial cells that promote lung fibrosis
Abstract
Background: Severe acute respiratory syndrome (SARS)-CoV-2-induced coronavirus disease-2019 (COVID-19) is a pandemic disease that affects > 2.8 million people worldwide, with numbers increasing dramatically daily. However, there is no specific treatment for COVID-19 and much remains unknown about this disease. Angiotensin-converting enzyme (ACE)2 is a cellular receptor of SARS-CoV-2. It is cleaved by type II transmembrane serine protease (TMPRSS)2 and disintegrin and metallopeptidase domain (ADAM)17 to assist viral entry into host cells. Clinically, SARS-CoV-2 infection may result in acute lung injury and lung fibrosis, but the underlying mechanisms of COVID-19 induced lung fibrosis are not fully understood.
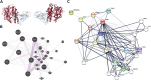
Methods: The networks of ACE2 and its interacting molecules were identified using bioinformatic methods. Their gene and protein expressions were measured in human epithelial cells after 24 h SARS-CoV-2 infection, or in existing datasets of lung fibrosis patients.
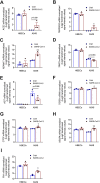
Results: We confirmed the binding of SARS-CoV-2 and ACE2 by bioinformatic analysis. TMPRSS2, ADAM17, tissue inhibitor of metalloproteinase (TIMP)3, angiotensinogen (AGT), transformation growth factor beta (TGFB1), connective tissue growth factor (CTGF), vascular endothelial growth factor (VEGF) A and fibronectin (FN) were interacted with ACE2, and the mRNA and protein of these molecules were expressed in lung epithelial cells. SARS-CoV-2 infection increased ACE2, TGFB1, CTGF and FN1 mRNA that were drivers of lung fibrosis. These changes were also found in lung tissues from lung fibrosis patients.
Conclusions: Therefore, SARS-CoV-2 binds with ACE2 and activates fibrosis-related genes and processes to induce lung fibrosis.
Keywords: Angiotensin-converting enzyme 2; Coronavirus; Lung fibrosis; SARS-CoV-2.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures





References
-
- WHO . Summary of probable SARS cases with onset of illness from 1 November 2002 to 31 July 2003. 2020.
-
- WHO . Epidemic and pandemic-prone diseases, MERS situation update, January 2020. 2020.
-
- WHO . Coronavirus disease 2019 (COVID-19) Situation Report – 125. 2020.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

