Herbivory and Attenuated UV Radiation Affect Volatile Emissions of the Invasive Weed Calluna vulgaris
- PMID: 32668802
- PMCID: PMC7397131
- DOI: 10.3390/molecules25143200
Herbivory and Attenuated UV Radiation Affect Volatile Emissions of the Invasive Weed Calluna vulgaris
Abstract
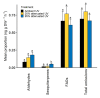
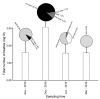
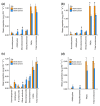
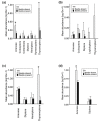
Calluna vulgaris (heather) is an aggressive invasive weed on the Central Plateau, North Is., New Zealand (NZ), where it encounters different environmental factors compared to its native range in Europe, such as high ultraviolet radiation (UV) and a lack of specialist herbivores. The specialist herbivore Lochmaea suturalis (heather beetle) was introduced from the United Kingdom (UK) in 1996 as a biocontrol agent to manage this invasive weed. Like other plant invaders, a novel environment may be challenging for heather as it adjusts to its new conditions. This process of "adjustment" involves morphological and physiological changes often linked to phenotypic plasticity. The biochemical responses of exotic plants to environmental variables in their invaded range is poorly understood. The production and release of volatile organic compounds (VOCs) is essential to plant communication and highly susceptible to environmental change. This study therefore aimed to explore the VOC emissions of heather in response to different levels of UV exposure, and to feeding damage by L. suturalis. Using tunnel houses clad with UV-selective filters, we measured VOCs produced by heather under NZ ambient, 20% attenuated, and 95% attenuated solar UV treatments. We also compared VOC emissions in the field at adjacent sites where L. suturalis was present or absent. Volatiles produced by the same target heather plants were measured at four different times in the spring and summer of 2018-2019, reflecting variations in beetle's abundance, feeding stage and plant phenology. Heather plants under 95% attenuated UV produced significantly higher amounts of (E)-β-farnesene, decanal, benzaldehyde, and benzeneacetaldehyde compared to 25% attenuated and ambient UV radiation. We also found significant differences in volatiles produced by heather plants in beetle-present versus beetle-absent sites on most sampling occasions. We also recorded a lower number of generalist herbivores on heather at sites where L. suturalis was present. Interactions between invasive plants, a novel environment, and the native communities they invade, are discussed.
Keywords: biocontrol agents; heather beetle; plant ecophysiology; plant secondary metabolites; plant volatiles; ultraviolet radiation; volatile organic compounds.
Conflict of interest statement
The authors declare no conflict of interests.
Figures


References
-
- Funk J.L. Differences in plasticity between invasive and native plants from a low resource environment. J. Ecol. 2008;96:1162–1173. doi: 10.1111/j.1365-2745.2008.01435.x. - DOI
-
- Keane R.M., Crawley M.J. Exotic plant invasions and the enemy release hypothesis. Trends Ecol. Evol. 2002;17:164–170. doi: 10.1016/S0169-5347(02)02499-0. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

