Experience with ospemifene in patients with vulvar and vaginal atrophy: case studies with bone marker profiles
- PMID: 32670383
- PMCID: PMC7337603
- DOI: 10.7573/dic.2020-3-3
Experience with ospemifene in patients with vulvar and vaginal atrophy: case studies with bone marker profiles
Abstract
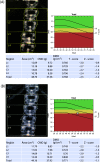
Selective estrogen receptor modulators (SERMs) exhibit varying agonist/antagonist activities on estrogen receptors in tissues, although most SERMs, including ospemifene, have agonist effects on bone. In this article, outcomes in relation to bone homeostasis, bone mineral density (BMD), and osteopenia-osteoporosis are examined in postmenopausal women during treatment with ospemifene for vulvar and vaginal atrophy (VVA), a component of the genitourinary syndrome of menopause. In cases 1 and 2, the women had established osteopenia or osteoporosis before the start of ospemifene treatment. After 6 months' and 7 months' treatment, respectively, marked reductions were observed in bone resorption (decreased levels of carboxy-terminal cross-linking telopeptide of type-1 collagen). The woman in case 3 had normal bone marker data and BMD prior to starting treatment with ospemifene. After 1 year, bone biomarkers and densitometry indicated improved bone health. Ospemifene 60 mg daily for treatment of VVA in postmenopausal women appears to benefit bone health although, because current evidence is based mainly on bone biomarkers, long-term studies are required to confirm this potential.
Keywords: bone biomarkers; bone homeostasis; ospemifene; vulvar and vaginal atrophy.
Copyright © 2020 Pingarrón Santofimia MC, González Rodríguez SP, Lilue M, Palacios S.
Conflict of interest statement
Disclosure and potential conflicts of interest: Dr. Pingarron reports grants and personal fees from Pfizer, grants and personal fees from MSD, grants and personal fees from Shionogi, grants and personal fees from Theramex, grants and personal fees from Exeltis, grants and personal fees from FAES, grants and personal fees from IPRAD, and grants and personal fees from Effik, outside the submitted work. Dr. Gonzalez Rodriguez reports grants from Pfizer, grants from Amgen, grants from Gedeon Ritcher, grants from Exeltis, grants from Bayer, grants from MSD, grants from Procare Health, grants and personal fees from Shionogi, grants from Serelys, and personal fees from Mylan, outside the submitted work. Dr. Lilue reports personal fees from Shionogi, outside the submitted work. Dr. Palacios reports grants from Pfizer, grants from Amgen, grants from Gedeon Ritcher, grants from Exeltis, grants from Bayer, grants from MSD, grants from Procare Health, grants and personal fees from Shionogi, grants from Serelys, and personal fees from Mylan, outside the submitted work. The authors have also provided scientific support to Shionogi Spain by lecturing and/or taking part in Advisory Board meetings organized by Shionogi (Madrid, Spain). The authors’ time was compensated by Shionogi Spain according to local codes of practice. The International Committee of Medical Journal Editors (ICMJE) Potential Conflicts of Interests form for the authors is available for download at: https://www.drugsincontext.com/wp-content/uploads/2020/05/dic.2020-3-3-COI.pdf
Figures





References
-
- Sullivan SD, Lehman A, Nathan NK, Thomson CA, Howard BV. Age of menopause and fracture risk in postmenopausal women randomized to calcium + vitamin D, hormone therapy, or the combination: results from the Women’s Health Initiative Clinical Trials. Menopause. 2017;24(4):371–378. doi: 10.1097/GME.0000000000000775. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

